Locus Coeruleus-Dorsolateral Septum Projections Modulate Depression-Like Behaviors via BDNF But Not Norepinephrine
- PMID: 38155473
- PMCID: PMC10933643
- DOI: 10.1002/advs.202303503
Locus Coeruleus-Dorsolateral Septum Projections Modulate Depression-Like Behaviors via BDNF But Not Norepinephrine
Abstract
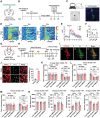
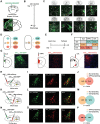
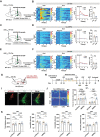
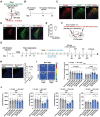
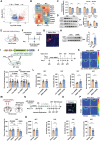
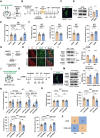
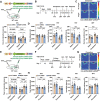
Locus coeruleus (LC) dysfunction is involved in the pathophysiology of depression; however, the neural circuits and specific molecular mechanisms responsible for this dysfunction remain unclear. Here, it is shown that activation of tyrosine hydroxylase (TH) neurons in the LC alleviates depression-like behaviors in susceptible mice. The dorsolateral septum (dLS) is the most physiologically relevant output from the LC under stress. Stimulation of the LCTH -dLSSST innervation with optogenetic and chemogenetic tools bidirectionally can regulate depression-like behaviors in both male and female mice. Mechanistically, it is found that brain-derived neurotrophic factor (BDNF), but not norepinephrine, is required for the circuit to produce antidepressant-like effects. Genetic overexpression of BDNF in the circuit or supplementation with BDNF protein in the dLS is sufficient to produce antidepressant-like effects. Furthermore, viral knockdown of BDNF in this circuit abolishes the antidepressant-like effect of ketamine, but not fluoxetine. Collectively, these findings underscore the notable antidepressant-like role of the LCTH -dLSSST pathway in depression via BDNF-TrkB signaling.
Keywords: BDNF signaling; depression-like behavior; dorsolateral septum; locus coeruleus.
© 2023 The Authors. Advanced Science published by Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Glucocorticoid receptor deletion from locus coeruleus norepinephrine neurons promotes depression-like social withdrawal in female but not male mice.Brain Res. 2019 May 1;1710:82-91. doi: 10.1016/j.brainres.2018.12.026. Epub 2018 Dec 18. Brain Res. 2019. PMID: 30576626
-
Stress Accelerates Defensive Responses to Looming in Mice and Involves a Locus Coeruleus-Superior Colliculus Projection.Curr Biol. 2018 Mar 19;28(6):859-871.e5. doi: 10.1016/j.cub.2018.02.005. Epub 2018 Mar 1. Curr Biol. 2018. PMID: 29502952
-
Saffron essential oil ameliorates CUMS-induced depression-like behavior in mice via the MAPK-CREB1-BDNF signaling pathway.J Ethnopharmacol. 2023 Jan 10;300:115719. doi: 10.1016/j.jep.2022.115719. Epub 2022 Sep 17. J Ethnopharmacol. 2023. PMID: 36126781
-
BDNF - a key transducer of antidepressant effects.Neuropharmacology. 2016 Mar;102:72-9. doi: 10.1016/j.neuropharm.2015.10.034. Epub 2015 Nov 11. Neuropharmacology. 2016. PMID: 26519901 Free PMC article. Review.
-
Causes, consequences, and cures for neuroinflammation mediated via the locus coeruleus: noradrenergic signaling system.J Neurochem. 2016 Oct;139 Suppl 2:154-178. doi: 10.1111/jnc.13447. Epub 2016 Mar 10. J Neurochem. 2016. PMID: 26968403 Review.
Cited by
-
Noradrenergic Locus Coeruleus-CA3 Activation Alleviates Neuropathic Pain and Anxiety- and Depression-Like Behaviors by Suppressing Microglial Neuroinflammation in SNI Mice.CNS Neurosci Ther. 2025 Mar;31(3):e70360. doi: 10.1111/cns.70360. CNS Neurosci Ther. 2025. PMID: 40130433 Free PMC article.
-
Unraveling the Pathogenesis of Post-Stroke Depression in a Hemorrhagic Mouse Model through Frontal Lobe Circuitry and JAK-STAT Signaling.Adv Sci (Weinh). 2024 Sep;11(33):e2402152. doi: 10.1002/advs.202402152. Epub 2024 Jul 1. Adv Sci (Weinh). 2024. PMID: 38946585 Free PMC article.
-
Visceral adipose tissue-derived extracellular vesicles promote stress susceptibility in obese mice via miR-140-5p.Acta Pharmacol Sin. 2025 May;46(5):1221-1235. doi: 10.1038/s41401-025-01484-z. Epub 2025 Feb 10. Acta Pharmacol Sin. 2025. PMID: 39930136
-
Regenerating Locus Coeruleus-Norepinephrine (LC-NE) Function: A Novel Approach for Neurodegenerative Diseases.Cell Prolif. 2025 Jun;58(6):e13807. doi: 10.1111/cpr.13807. Epub 2025 Jan 28. Cell Prolif. 2025. PMID: 39876531 Free PMC article. Review.
-
A molecularly distinct cell type in the midbrain regulates intermale aggression behaviors in mice.Theranostics. 2025 Jan 1;15(2):707-725. doi: 10.7150/thno.101658. eCollection 2025. Theranostics. 2025. PMID: 39744695 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
