Development of hydrogel-based standards and phantoms for non-linear imaging at depth
- PMID: 38155703
- PMCID: PMC10753126
- DOI: 10.1117/1.JBO.28.12.126007
Development of hydrogel-based standards and phantoms for non-linear imaging at depth
Abstract
Significance: Rapid advances in medical imaging technology, particularly the development of optical systems with non-linear imaging modalities, are boosting deep tissue imaging. The development of reliable standards and phantoms is critical for validation and optimization of these cutting-edge imaging techniques.
Aim: We aim to design and fabricate flexible, multi-layered hydrogel-based optical standards and evaluate advanced optical imaging techniques at depth.
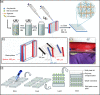
Approach: Standards were made using a robust double-network hydrogel matrix consisting of agarose and polyacrylamide. The materials generated ranged from single layers to more complex constructs consisting of up to seven layers, with modality-specific markers embedded between the layers.
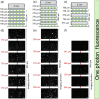
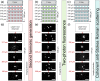
Results: These standards proved useful in the determination of the axial scaling factor for light microscopy and allowed for depth evaluation for different imaging modalities (conventional one-photon excitation fluorescence imaging, two-photon excitation fluorescence imaging, second harmonic generation imaging, and coherent anti-Stokes Raman scattering) achieving actual depths of 1550, 1550, 1240, and , respectively. Once fabricated, the phantoms were found to be stable for many months.
Conclusions: The ability to image at depth, the phantom's robustness and flexible layered structure, and the ready incorporation of "optical markers" make these ideal depth standards for the validation of a variety of imaging modalities.
Keywords: axial scaling; depth imaging; hydrogel; non-linear imaging; phantoms; standards.
© 2023 The Authors.
Figures



References
-
- Kasban H., El-Bendary M., Salama D., “A comparative study of medical imaging techniques,” Int. J. Inf. Sci. Intell. Syst. 4(2), 37–58 (2015).

