Propidium monoazide PCR, a method to determine OsHV-1 undamaged capsids and to estimate virus Lethal Dose 50
- PMID: 38160910
- PMCID: PMC10800765
- DOI: 10.1016/j.virusres.2023.199307
Propidium monoazide PCR, a method to determine OsHV-1 undamaged capsids and to estimate virus Lethal Dose 50
Abstract
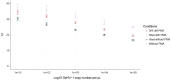
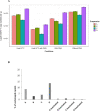
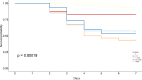
Ostreid herpes virus 1 (OsHV-1) has been classified within the Malacoherpesviridae family from the Herpesvirales order. OsHV-1 is the etiological agent of a contagious viral disease of Pacific oysters, C. gigas, affecting also other bivalve species. Mortality rates reported associated with the viral infection vary considerably between sites and countries and depend on the age of affected stocks. A variant called μVar has been reported since 2008 in Europe and other variants in Australia and in New Zealand last decade. These variants are considered as the main causative agents of mass mortality events affecting C. gigas. Presently there is no established cell line that allows for the detection of infectious OsHV-1. In this context, a technique of propidium monoazide (PMA) PCR was developed in order to quantify "undamaged" capsids. This methodology is of interest to explore the virus infectivity. Being able to quantify viral particles getting an undamaged capsid (not only an amount of viral DNA) in tissue homogenates prepared from infected oysters or in seawater samples can assist in the definition of a Lethal Dose (LD) 50 and gain information in the experiments conducted to reproduce the viral infection. The main objectives of the present study were (i) the development/optimization of a PMA PCR technique for OsHV-1 detection using the best quantity of PMA and verifying its effectiveness through heat treatment, (ii) the definition of the percentage of undamaged capsids in four different tissue homogenates prepared from infected Pacific oysters and (iii) the approach of a LD50 during experimental viral infection assays on the basis of a number of undamaged capsids. Although the developped PMA PCR technique was unable to determine OsHV-1 infectivity in viral supensions, it could greatly improve interpretation of virus positive results obtained by qPCR. This technique is not intended to replace the quantification of viral DNA by qPCR, but it does make it possible to give a form of biological meaning to the detection of this DNA.
Keywords: Lethal Dose 50; Mortality; OsHV-1; PCR; Pacific oyster; Propidium monoazide; Undamaged capsids.
Copyright © 2023. Published by Elsevier B.V.
Conflict of interest statement
Declaration of Competing Interest The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures

References
-
- Azéma P., Travers M.A., Benabdelmouna A., Dégremont L. Single or dual experimental infections with Vibrio aestuarianus and OsHV-1 in diploid and triploid Crassostrea gigas at the spat, juvenile and adult stages. J. Pathol. 2016;139:92–101. - PubMed
-
- Canh V.D., Liu M., Sangsanont J., Katayama H. Capsid integrity detection of pathogenic viruses in waters: recent progress and potential furture applications. Sci. Total Environ. 2022;827:154258–154272. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

