A desmosomal cadherin controls multipotent hair follicle stem cell quiescence and orchestrates regeneration through adhesion signaling
- PMID: 38162019
- PMCID: PMC10755723
- DOI: 10.1016/j.isci.2023.108568
A desmosomal cadherin controls multipotent hair follicle stem cell quiescence and orchestrates regeneration through adhesion signaling
Abstract
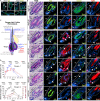
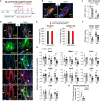
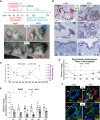
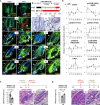
Stem cells (SCs) are critical to maintain tissue homeostasis. However, it is currently not known whether signaling through cell junctions protects quiescent epithelial SC reservoirs from depletion during disease-inflicted damage. Using the autoimmune model disease pemphigus vulgaris (PV), this study reveals an unprecedented role for a desmosomal cadherin in governing SC quiescence and regeneration through adhesion signaling in the multipotent mouse hair follicle compartment known as the bulge. Autoantibody-mediated, mechanical uncoupling of desmoglein (Dsg) 3 transadhesion activates quiescent bulge SC which lose their multipotency and stemness, become actively cycling, and finally delaminate from their epithelial niche. This then initiates a self-organized regenerative program which restores Dsg3 function and bulge morphology including SC quiescence and multipotency. These profound changes are triggered by the sole loss of functional Dsg3, resemble major signaling events in Dsg3-/- mice, and are driven by SC-relevant EGFR activation and Wnt modulation requiring longitudinal repression of Hedgehog signaling.
Keywords: Cell biology; Molecular biology.
© 2023 The Author(s).
Conflict of interest statement
The last author (E.J.M.) is a founder and director of the board of CELLnTEC Advanced Cell Systems AG. We used CELLnTEC media in this study. All other authors have declared no conflict of interest.
Figures


References
-
- Wagers A.J. The stem cell niche in regenerative medicine. Cell Stem Cell. 2012;10:362–369. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

