Enzymatic Assembly of Diverse Lactone Structures: An Intramolecular C-H Functionalization Strategy
- PMID: 38166100
- PMCID: PMC11290351
- DOI: 10.1021/jacs.3c11722
Enzymatic Assembly of Diverse Lactone Structures: An Intramolecular C-H Functionalization Strategy
Abstract
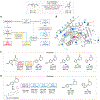
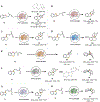
Lactones are cyclic esters with extensive applications in materials science, medicinal chemistry, and the food and perfume industries. Nature's strategy for the synthesis of many lactones found in natural products always relies on a single type of retrosynthetic strategy, a C-O bond disconnection. Here, we describe a set of laboratory-engineered enzymes that use a new-to-nature C-C bond-forming strategy to assemble diverse lactone structures. These engineered "carbene transferases" catalyze intramolecular carbene insertions into benzylic or allylic C-H bonds, which allow for the synthesis of lactones with different ring sizes and ring scaffolds from simple starting materials. Starting from a serine-ligated cytochrome P450 variant previously engineered for other carbene-transfer activities, directed evolution generated a variant P411-LAS-5247, which exhibits a high activity for constructing a five-membered ε-lactone, lactam, and cyclic ketone products (up to 5600 total turnovers (TTN) and >99% enantiomeric excess (ee)). Further engineering led to variants P411-LAS-5249 and P411-LAS-5264, which deliver six-membered δ-lactones and seven-membered ε-lactones, respectively, overcoming the thermodynamically unfavorable ring strain associated with these products compared to the γ-lactones. This new carbene-transfer activity was further extended to the synthesis of complex lactone scaffolds based on fused, bridged, and spiro rings. The enzymatic platform developed here complements natural biosynthetic strategies for lactone assembly and expands the structural diversity of lactones accessible through C-H functionalization.
Figures


Similar articles
-
Engineered Cytochrome c-Catalyzed Lactone-Carbene B-H Insertion.Synlett. 2019 Mar;30(4):378-382. doi: 10.1055/s-0037-1611662. Epub 2019 Jan 14. Synlett. 2019. PMID: 30930550 Free PMC article.
-
An Enzymatic Platform for the Highly Enantioselective and Stereodivergent Construction of Cyclopropyl-δ-lactones.Angew Chem Int Ed Engl. 2020 Nov 23;59(48):21634-21639. doi: 10.1002/anie.202007953. Epub 2020 Sep 17. Angew Chem Int Ed Engl. 2020. PMID: 32667122 Free PMC article.
-
Engineering Cytochrome P450s for Enantioselective Cyclopropenation of Internal Alkynes.J Am Chem Soc. 2020 Apr 15;142(15):6891-6895. doi: 10.1021/jacs.0c01313. Epub 2020 Mar 31. J Am Chem Soc. 2020. PMID: 32223130
-
Biosynthesis and chemical diversity of β-lactone natural products.Nat Prod Rep. 2019 Mar 20;36(3):458-475. doi: 10.1039/c8np00052b. Nat Prod Rep. 2019. PMID: 30191940 Review.
-
Navigating the Unnatural Reaction Space: Directed Evolution of Heme Proteins for Selective Carbene and Nitrene Transfer.Acc Chem Res. 2021 Mar 2;54(5):1209-1225. doi: 10.1021/acs.accounts.0c00591. Epub 2021 Jan 25. Acc Chem Res. 2021. PMID: 33491448 Free PMC article. Review.
Cited by
-
C 4-Symmetric Bowl-Shaped Diruthenium Tetracarboxylate Catalysts for Enantioselective C-H Functionalization Using Donor/Acceptor Carbenes.ACS Catal. 2025 Mar 27;15(8):5906-5914. doi: 10.1021/acscatal.5c01052. eCollection 2025 Apr 18. ACS Catal. 2025. PMID: 40270881 Free PMC article.
-
Catalyzing the future: recent advances in chemical synthesis using enzymes.Curr Opin Chem Biol. 2024 Dec;83:102536. doi: 10.1016/j.cbpa.2024.102536. Epub 2024 Oct 5. Curr Opin Chem Biol. 2024. PMID: 39369557 Free PMC article. Review.
-
Amines tuned controllable carbonylation for the synthesis of γ-lactones and 1,4-diones.Nat Commun. 2025 Jul 8;16(1):6305. doi: 10.1038/s41467-025-61762-6. Nat Commun. 2025. PMID: 40628753 Free PMC article.
References
-
- Sartori SK; Diaz MAN; Diaz-Muñoz G. Lactones: Classification, Synthesis, Biological Activities, and Industrial Applications. Tetrahedron 2021, 84, 132001.
-
- Doyle MP; Forbes DC Recent Advances in Asymmetric Catalytic Metal Carbene Transformations. Chem. Rev. 1998, 98, 911–936. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

