Hijacking of internal calcium dynamics by intracellularly residing viral rhodopsins
- PMID: 38167346
- PMCID: PMC10761956
- DOI: 10.1038/s41467-023-44548-6
Hijacking of internal calcium dynamics by intracellularly residing viral rhodopsins
Abstract
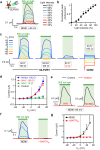
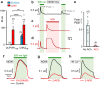
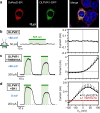
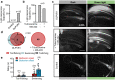
Rhodopsins are ubiquitous light-driven membrane proteins with diverse functions, including ion transport. Widely distributed, they are also coded in the genomes of giant viruses infecting phytoplankton where their function is not settled. Here, we examine the properties of OLPVR1 (Organic Lake Phycodnavirus Rhodopsin) and two other type 1 viral channelrhodopsins (VCR1s), and demonstrate that VCR1s accumulate exclusively intracellularly, and, upon illumination, induce calcium release from intracellular IP3-dependent stores. In vivo, this light-induced calcium release is sufficient to remote control muscle contraction in VCR1-expressing tadpoles. VCR1s natively confer light-induced Ca2+ release, suggesting a distinct mechanism for reshaping the response to light of virus-infected algae. The ability of VCR1s to photorelease calcium without altering plasma membrane electrical properties marks them as potential precursors for optogenetics tools, with potential applications in basic research and medicine.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures

Similar articles
-
Viral rhodopsins 1 are an unique family of light-gated cation channels.Nat Commun. 2020 Nov 11;11(1):5707. doi: 10.1038/s41467-020-19457-7. Nat Commun. 2020. PMID: 33177509 Free PMC article.
-
History and Perspectives of Ion-Transporting Rhodopsins.Adv Exp Med Biol. 2021;1293:3-19. doi: 10.1007/978-981-15-8763-4_1. Adv Exp Med Biol. 2021. PMID: 33398804 Review.
-
[How to Choose the Best Optogenetic Tool for Your Research].Brain Nerve. 2024 Jul;76(7):835-842. doi: 10.11477/mf.1416202691. Brain Nerve. 2024. PMID: 38970320 Review. Japanese.
-
Advances and prospects of rhodopsin-based optogenetics in plant research.Plant Physiol. 2021 Oct 5;187(2):572-589. doi: 10.1093/plphys/kiab338. Plant Physiol. 2021. PMID: 35237820 Free PMC article. Review.
-
Novel pH-Sensitive Microbial Rhodopsin from Sphingomonas paucimobilis.Dokl Biochem Biophys. 2020 Nov;495(1):342-346. doi: 10.1134/S1607672920060162. Epub 2020 Dec 25. Dokl Biochem Biophys. 2020. PMID: 33368048
Cited by
-
Optogenetic engineering for ion channel modulation.Curr Opin Chem Biol. 2025 Apr;85:102569. doi: 10.1016/j.cbpa.2025.102569. Epub 2025 Feb 3. Curr Opin Chem Biol. 2025. PMID: 39903997 Review.
-
Ion-conducting and gating molecular mechanisms of channelrhodopsin revealed by true-atomic-resolution structures of open and closed states.Nat Struct Mol Biol. 2025 Aug;32(8):1347-1357. doi: 10.1038/s41594-025-01488-7. Epub 2025 Apr 9. Nat Struct Mol Biol. 2025. PMID: 40205223
-
Combined Mutational and Spectroscopic Study on the Calcium-Related Kinetic Effects on the VirChR1 Photocycle.J Phys Chem B. 2025 Mar 20;129(11):2946-2957. doi: 10.1021/acs.jpcb.4c08416. Epub 2025 Mar 10. J Phys Chem B. 2025. PMID: 40063977 Free PMC article.
-
Spatiotemporal dynamics of giant viruses within a deep freshwater lake reveal a distinct dark-water community.ISME J. 2024 Jan 8;18(1):wrae182. doi: 10.1093/ismejo/wrae182. ISME J. 2024. PMID: 39312489 Free PMC article.
-
Theoretical analysis of low power optogenetic control of synaptic plasticity with subcellular expression of CapChR2 at postsynaptic spine.Sci Rep. 2025 Apr 1;15(1):11166. doi: 10.1038/s41598-025-95355-6. Sci Rep. 2025. PMID: 40169824 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

