The fluorescent ligand bTVBT2 reveals increased p-tau uptake by retinal microglia in Alzheimer's disease patients and AppNL-F/NL-F mice
- PMID: 38167557
- PMCID: PMC10763304
- DOI: 10.1186/s13195-023-01375-7
The fluorescent ligand bTVBT2 reveals increased p-tau uptake by retinal microglia in Alzheimer's disease patients and AppNL-F/NL-F mice
Abstract
Background: Amyloid beta (Aβ) deposits and hyperphosphorylated tau (p-tau) accumulation have been identified in the retina of Alzheimer's disease (AD) patients and transgenic AD mice. Previous studies have shown that retinal microglia engulf Aβ, but this property decreases in AD patients. Whether retinal microglia also take up p-tau and if this event is affected in AD is yet not described. In the current study, we use the p-tau-specific thiophene-based ligand bTVBT2 to investigate the relationship between disease progression and p-tau uptake by microglia in the retina of AD patients and AppNL-F/NL-F knock-in mice, an AD mouse model known to demonstrate extracellular Aβ plaques and dystrophic neurites in the brain from 6 months of age.
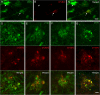
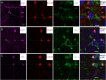
Methods: Evaluation of bTVBT2 specificity and its presence within microglia was assessed by immunofluorescent staining of hippocampal sections and flat-mount retina samples from non-demented controls, AD patients, 3-, 9-, and 12-month-old AppNL-F/NL-F knock-in mice and 12- and 18-month-old wild type (WT) mice. We used ImageJ to analyze the amount of bTVBT2 inside Iba1-positive microglia. Co-localization between the ligand and p-tau variant Ser396/Ser404 (PHF-1), Aβ, phosphorylated TAR DNA binding protein 43 (pTDP-43), and islet amyloid polypeptide (IAPP) in the brain and retina was analyzed using confocal imaging.
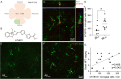
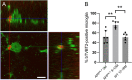
Results: Confocal imaging analysis showed that bTVBT2 binds to PHF-1- and AT8-positive aggregates inside retinal microglia, and not to Aβ, pTDP-43, or IAPP. The density of bTVBT2-positive microglia was higher in cases with a high Aβ load compared to those with a low Aβ load. This density correlated with the neurofibrillary tangle load in the brain, but not with retinal levels of high molecular weight (aggregated) Aβ40 or Aβ42. Analysis of AppNL-F/NL-F knock-in mouse retina further showed that 50% of microglia in 3-month-old AppNL-F/NL-F knock-in mice contained bTVBT2. The percentage significantly increased in 9- and 12-month-old mice.
Conclusion: Our study suggests that the microglial capability to uptake p-tau in the retina persists and intensifies with AD progression. These results also highlight bTVBT2 as a ligand of interest in future monitoring of retinal AD pathology.
Keywords: Alzheimer’s disease; Retina; Tauopathy; Thiophene-based ligands.
© 2023. The Author(s).
Conflict of interest statement
M.W. has acquired research support (for the institution) from Eli Lilly. OH has acquired research support (for the institution) from ADx, AVID Radiopharmaceuticals, Biogen, Eli Lilly, Eisai, Fujirebio, GE Healthcare, Pfizer, and Roche. In the past 2 years, he has received consultancy/speaker fees from AC Immune, Amylyx, Alzpath, BioArctic, Biogen, Cerveau, Eisai, Eli Lilly, Fujirebio, Merck, Novartis, Novo Nordisk, Roche, Sanofi and Siemens.
Figures
Similar articles
-
Plaque associated microglia hyper-secrete extracellular vesicles and accelerate tau propagation in a humanized APP mouse model.Mol Neurodegener. 2021 Mar 22;16(1):18. doi: 10.1186/s13024-021-00440-9. Mol Neurodegener. 2021. PMID: 33752701 Free PMC article.
-
Increase in Tau Pathology in P290S Mapt Knock-In Mice Crossed with App NL-G-F Mice.eNeuro. 2022 Dec 20;9(6):ENEURO.0247-22.2022. doi: 10.1523/ENEURO.0247-22.2022. Print 2022 Nov-Dec. eNeuro. 2022. PMID: 36635241 Free PMC article.
-
Amyloid-β plaque formation and reactive gliosis are required for induction of cognitive deficits in App knock-in mouse models of Alzheimer's disease.BMC Neurosci. 2019 Mar 20;20(1):13. doi: 10.1186/s12868-019-0496-6. BMC Neurosci. 2019. PMID: 30894120 Free PMC article.
-
Effects of CX3CR1 and Fractalkine Chemokines in Amyloid Beta Clearance and p-Tau Accumulation in Alzheimer's Disease (AD) Rodent Models: Is Fractalkine a Systemic Biomarker for AD?Curr Alzheimer Res. 2016;13(4):403-12. doi: 10.2174/1567205013666151116125714. Curr Alzheimer Res. 2016. PMID: 26567742 Review.
-
Recent Advances in the Modeling of Alzheimer's Disease.Front Neurosci. 2022 Mar 31;16:807473. doi: 10.3389/fnins.2022.807473. eCollection 2022. Front Neurosci. 2022. PMID: 35431779 Free PMC article. Review.
Cited by
-
Identification of Chlamydia pneumoniae and NLRP3 inflammasome activation in Alzheimer's disease retina.Res Sq [Preprint]. 2025 Jun 26:rs.3.rs-6658954. doi: 10.21203/rs.3.rs-6658954/v1. Res Sq. 2025. PMID: 40678205 Free PMC article. Preprint.
-
Identification of retinal oligomeric, citrullinated, and other tau isoforms in early and advanced AD and relations to disease status.Acta Neuropathol. 2024 Jul 9;148(1):3. doi: 10.1007/s00401-024-02760-8. Acta Neuropathol. 2024. PMID: 38980423 Free PMC article.
-
Alzheimer's disease pathophysiology in the Retina.Prog Retin Eye Res. 2024 Jul;101:101273. doi: 10.1016/j.preteyeres.2024.101273. Epub 2024 May 15. Prog Retin Eye Res. 2024. PMID: 38759947 Free PMC article. Review.
-
Identification of Chlamydia pneumoniae and NLRP3 inflammasome activation in Alzheimer's disease retina.bioRxiv [Preprint]. 2025 Jun 25:2025.06.19.660619. doi: 10.1101/2025.06.19.660619. bioRxiv. 2025. PMID: 40667156 Free PMC article. Preprint.
-
Retinal Vascular and Structural Changes in the Murine Alzheimer's APPNL-F/NL-F Model from 6 to 20 Months.Biomolecules. 2024 Jul 10;14(7):828. doi: 10.3390/biom14070828. Biomolecules. 2024. PMID: 39062542 Free PMC article.
References
-
- Mirra SS, Heyman A, McKeel D, Sumi SM, Crain BJ, Brownlee LM, et al. The Consortium to Establish a Registry for Alzheimer's Disease (CERAD). Part II. Standardization of the neuropathologic assessment of Alzheimer's disease. Neurology. 1991;41(4):479–86. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

