This is a preprint.
Clinical utility of plasma Aβ42/40 ratio by LC-MS/MS in Alzheimer's disease assessment
- PMID: 38168329
- PMCID: PMC10760303
- DOI: 10.1101/2023.12.12.23299878
Clinical utility of plasma Aβ42/40 ratio by LC-MS/MS in Alzheimer's disease assessment
Update in
-
Clinical utility of plasma Aβ42/40 ratio by LC-MS/MS in Alzheimer's disease assessment.Front Neurol. 2024 Mar 25;15:1364658. doi: 10.3389/fneur.2024.1364658. eCollection 2024. Front Neurol. 2024. PMID: 38595851 Free PMC article.
Abstract
Introduction: Plasma Aβ42/40 ratio can be used to help predict amyloid PET status, but its clinical utility in Alzheimer's disease (AD) assessment is unclear.
Methods: Aβ42/40 ratio was measured by LC-MS/MS in 250 specimens with associated amyloid PET imaging, diagnosis, and demographic data, and 6,192 consecutive clinical specimens submitted for Aβ42/40 testing.
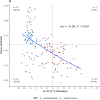
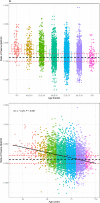
Results: High diagnostic sensitivity and negative predictive value (NPV) for Aβ-PET positivity were observed, consistent with the clinical performance of other plasma LC-MS/MS assays, but with greater separation between Aβ42/40 values for individuals with positive vs negative Aβ-PET results. Assuming a moderate prevalence of Aβ-PET positivity, a cutpoint was identified with 99% NPV, which could help predict that AD is likely not the cause of patients' cognitive impairment and help reduce PET evaluation by about 40%.
Discussion: Using high-throughput plasma Aβ42/40 LC-MS/MS assays can help reduce PET evaluations in patients with low likelihood of AD pathology, allowing for cost savings.
Keywords: Alzheimer’s disease; LC-MS/MS; PET; amyloid; biomarkers; blood biomarkers; prescreening.
Conflict of interest statement
CONFLICT OF INTEREST STATEMENT Darren M. Weber, Steven W. Taylor, Robert J. Lagier, Jueun C. Kim, Scott M. Goldman, Nigel J. Clarke, and Michael K. Racke are employees of Quest Diagnostics. All other authors have nothing to disclose.
Figures







References
-
- Lane CA, Hardy J, Schott JM. Alzheimer’s disease. Eur J Neurol. 2018;25:59–70. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
