Prodigiosin/celecoxib-loaded into zein/sodium caseinate nanoparticles as a potential therapy for triple negative breast cancer
- PMID: 38168547
- PMCID: PMC10761898
- DOI: 10.1038/s41598-023-50531-4
Prodigiosin/celecoxib-loaded into zein/sodium caseinate nanoparticles as a potential therapy for triple negative breast cancer
Abstract
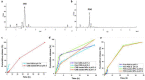
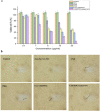
Nowadays, breast cancer is considered one of the most upsetting malignancies among females. Encapsulation of celecoxib (CXB) and prodigiosin (PDG) into zein/sodium caseinate nanoparticles (NPs) produce homogenous and spherical nanoparticles with good encapsulation efficiencies (EE %) and bioavailability. In vitro cytotoxicity study conducted on human breast cancer MDA-MB-231 cell lines revealed that there was a significant decline in the IC50 for encapsulated drugs when compared to each drug alone or their free combination. In addition, results demonstrated that there is a synergism between CXB and PDG as their combination indices were 0.62251 and 0.15493, respectively. Moreover, results of scratch wound healing assay revealed enhanced antimigratory effect of free drugs and fabricated NPs in comparison to untreated cells. Furthermore, In vitro results manifested that formulated nanoparticles exhibited induction of apoptosis associated with reduced angiogenesis, proliferation, and inflammation. In conclusion, nanoencapsulation of multiple drugs into nanoparticles might be a promising approach to develop new therapies for the managing of triple negative breast cancer.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures







Similar articles
-
Fabrication of ibuprofen/naringenin-coloaded into zein/sodium caseinate nanoparticles: evaluation of antiproliferative activity and apoptosis induction in liver cancer cells.J Biomater Sci Polym Ed. 2024 Dec;35(17):2703-2722. doi: 10.1080/09205063.2024.2391653. Epub 2024 Sep 1. J Biomater Sci Polym Ed. 2024. PMID: 39217620
-
One-step assembly of zein/caseinate/alginate nanoparticles for encapsulation and improved bioaccessibility of propolis.Food Funct. 2019 Feb 20;10(2):635-645. doi: 10.1039/c8fo01614c. Food Funct. 2019. PMID: 30648709
-
Fabrication and characterization of teriflunomide-loaded chondroitin sulfate hybridized zein nanoparticles for the management of triple negative breast cancer.Int J Biol Macromol. 2025 Apr;300:140316. doi: 10.1016/j.ijbiomac.2025.140316. Epub 2025 Jan 24. Int J Biol Macromol. 2025. PMID: 39864685
-
Zein-sodium caseinate-diosmin nanoparticles as a promising anti-cancer agent with targeted efficacy against A2780 cell line.Sci Rep. 2025 Mar 13;15(1):8762. doi: 10.1038/s41598-025-93772-1. Sci Rep. 2025. PMID: 40082691 Free PMC article.
-
Enhancement of lemongrass essential oil physicochemical properties and antibacterial activity by encapsulation in zein-caseinate nanocomposite.Sci Rep. 2024 Jul 27;14(1):17278. doi: 10.1038/s41598-024-67273-6. Sci Rep. 2024. PMID: 39068244 Free PMC article.
Cited by
-
Research advances in Zein-based nano-delivery systems.Front Nutr. 2024 May 9;11:1379982. doi: 10.3389/fnut.2024.1379982. eCollection 2024. Front Nutr. 2024. PMID: 38798768 Free PMC article. Review.
-
Evaluation of the Combinatory Anticancer Effect of Chemotherapeutic Compounds and Prodigiosin against HCT-116, LoVo, and A549 Cell lines.ACS Omega. 2024 Nov 26;9(49):48112-48124. doi: 10.1021/acsomega.4c04760. eCollection 2024 Dec 10. ACS Omega. 2024. PMID: 39676943 Free PMC article.
References
-
- Chopra S, Davies EL. Breast cancer. Med. U. K. 2020;2020:113–118. doi: 10.1016/j.mpmed.2019.11.009. - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

