Cerebrospinal fluid immunological cytokines predict intracranial tumor response to immunotherapy in non-small cell lung cancer patients with brain metastases
- PMID: 38169917
- PMCID: PMC10761018
- DOI: 10.1080/2162402X.2023.2290790
Cerebrospinal fluid immunological cytokines predict intracranial tumor response to immunotherapy in non-small cell lung cancer patients with brain metastases
Abstract
Background: Immunotherapy has shown intracranial efficacy in non-small cell lung cancer (NSCLC) patients with brain metastases. However, predictive biomarkers for intracranial response to immunotherapy are lacking. This post-hoc analysis aimed to explore the potential of immunological cytokines in cerebrospinal fluid (CSF) to predict intracranial tumor response to immunotherapy in patients with brain metastases.
Methods: Treatment-naive NSCLC patients with brain metastases who received camrelizumab plus chemotherapy were enrolled. Paired plasma and CSF samples were prospectively collected at baseline and the first treatment assessment. All samples were analyzed for 92 immuno-oncology cytokines using Olink's panels.
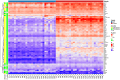
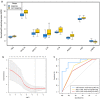
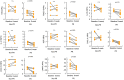
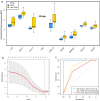
Results: A total of 28 patients were included in this analysis. At baseline, most immunological cytokines were significantly lower in CSF than in plasma, whereas a subset comprising CD83, PTN, TNFRSF21, TWEAK, ICOSLG, DCN, IL-8, and MCP-1, was increased in CSF. Baseline CSF levels of LAMP3 were significantly higher in patients with intracranial tumor response, while the levels of CXCL10, IL-12, CXCL11, IL-18, TIE2, HGF, and PDCD1 were significantly lower. Furthermore, the CXCL10, CXCL11, TIE2, PDCD1, IL-18, HGF, and LAMP3 in CSF were also significantly associated with intracranial progression-free survival for immunotherapy. The identified cytokines in CSF were decreased at the first treatment evaluation in patients with intracranial tumor response. The logistic CSF immuno-cytokine model yielded an AUC of 0.91, as compared to PD-L1 expression (AUC of 0.72).
Conclusions: Immunological cytokines in CSF could predict intracranial tumor response to immunotherapy in NSCLC patients with brain metastases, and the findings warrant validation in a larger prospective cohort study.
Trial registration: ClinicalTrials.gov identifier: NCT04211090.
Keywords: Brain metastases; cerebrospinal fluid; immunological cytokines; immunotherapy; tumor response.
© 2023 The Author(s). Published with license by Taylor & Francis Group, LLC.
Conflict of interest statement
No potential conflict of interest was reported by the authors.
Figures
Similar articles
-
Systemic and Intracranial Outcomes With First-Line Nivolumab Plus Ipilimumab in Patients With Metastatic NSCLC and Baseline Brain Metastases From CheckMate 227 Part 1.J Thorac Oncol. 2023 Aug;18(8):1055-1069. doi: 10.1016/j.jtho.2023.04.021. Epub 2023 May 3. J Thorac Oncol. 2023. PMID: 37146754 Clinical Trial.
-
Utilizing phenotypic characteristics of metastatic brain tumors to improve the probability of detecting circulating tumor DNA from cerebrospinal fluid in non-small-cell lung cancer patients: development and validation of a prediction model in a prospective cohort study.ESMO Open. 2022 Feb;7(1):100305. doi: 10.1016/j.esmoop.2021.100305. Epub 2021 Dec 15. ESMO Open. 2022. PMID: 34922300 Free PMC article.
-
Anti-PD-(L)1 immunotherapy for brain metastases in non-small cell lung cancer: Mechanisms, advances, and challenges.Cancer Lett. 2021 Apr 1;502:166-179. doi: 10.1016/j.canlet.2020.12.043. Epub 2021 Jan 13. Cancer Lett. 2021. PMID: 33450361
-
Immunotherapy in NSCLC patients with brain metastases. Understanding brain tumor microenvironment and dissecting outcomes from immune checkpoint blockade in the clinic.Cancer Treat Rev. 2020 Sep;89:102067. doi: 10.1016/j.ctrv.2020.102067. Epub 2020 Jul 7. Cancer Treat Rev. 2020. PMID: 32682248 Review.
-
Predicting the efficacy of first-line immunotherapy by combining cancer cachexia and tumor burden in advanced non-small cell lung cancer.Thorac Cancer. 2022 Jul;13(14):2064-2074. doi: 10.1111/1759-7714.14529. Epub 2022 Jun 13. Thorac Cancer. 2022. PMID: 35698259 Free PMC article. Review.
Cited by
-
Immunotherapy in lung cancer brain metastases.NPJ Precis Oncol. 2025 May 6;9(1):130. doi: 10.1038/s41698-025-00901-0. NPJ Precis Oncol. 2025. PMID: 40328894 Free PMC article. Review.
-
Efficacy and safety of immune checkpoint inhibitors for brain metastases of non-small cell lung cancer: a systematic review and network meta-analysis.Front Oncol. 2025 Apr 16;15:1513774. doi: 10.3389/fonc.2025.1513774. eCollection 2025. Front Oncol. 2025. PMID: 40308503 Free PMC article.
-
Clinical and Translational Results from PORTER, a Multicohort Phase I Platform Trial of Combination Immunotherapy in Metastatic Castration-Resistant Prostate Cancer.Clin Cancer Res. 2025 Apr 14;31(8):1463-1475. doi: 10.1158/1078-0432.CCR-24-3693. Clin Cancer Res. 2025. PMID: 39964352 Free PMC article. Clinical Trial.
References
-
- Tabouret E, Chinot O, Metellus P, Tallet A, Viens P, Gonçalves A.. Recent trends in epidemiology of brain metastases: an overview. Anticancer Res. 2012;32:4655–11. - PubMed
-
- Goldberg SB, Schalper KA, Gettinger SN, Mahajan A, Herbst RS, Chiang AC, Lilenbaum R, Wilson FH, Omay SB, Yu JB, et al. Pembrolizumab for management of patients with NSCLC and brain metastases: long-term results and biomarker analysis from a non-randomised, open-label, phase 2 trial. Lancet Oncol. 2020;21(5):655–663. doi:10.1016/S1470-2045(20)30111-X. - DOI - PMC - PubMed
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous
