Light-enhanced VEGF121/rGel induce immunogenic cell death and increase the antitumor activity of αCTLA4 treatment
- PMID: 38173721
- PMCID: PMC10762878
- DOI: 10.3389/fimmu.2023.1278000
Light-enhanced VEGF121/rGel induce immunogenic cell death and increase the antitumor activity of αCTLA4 treatment
Erratum in
-
Corrigendum: Light-enhanced VEGF121/rGel induce immunogenic cell death and increase the antitumor activity of αCTLA4 treatment.Front Immunol. 2024 Jan 9;14:1359973. doi: 10.3389/fimmu.2023.1359973. eCollection 2023. Front Immunol. 2024. PMID: 38264651 Free PMC article.
Abstract
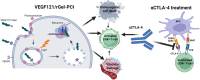
Background: Immune-checkpoint inhibitors (ICIs) represent a revolution in cancer therapy and are currently implemented as standard therapy within several cancer indications. Nevertheless, the treatment is only effective in a subset of patients, and immune-related adverse effects complicate the improved survival. Adjuvant treatments that can improve the efficacy of ICIs are highly warranted, not only to increase the response rate, but also to reduce the therapeutic ICI dosage. Several treatment modalities have been suggested as ICI adjuvants including vascular targeted treatments and photodynamic therapy (PDT). Photochemical internalization (PCI) is a drug delivery system, based on PDT. PCI is long known to generate an immune response in murine models and was recently shown to enhance the cellular immune response of a vaccine in a clinical study. In the present work we evaluated PCI in combination with the vascular targeting toxin VEGF121/rGel with respect to induction of immune-mediated cell death as well as in vitro ICI enhancement.
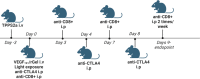
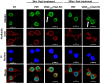
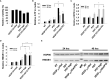
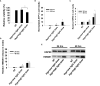
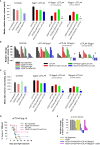
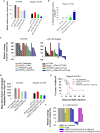
Methods: DAMP signaling post VEGF121/rGel-PCI was assessed in CT26 and MC38 murine colon cancer cell lines. Hypericin-PDT, previously indicated as an highly efficient DAMP inducer (but difficult to utilize clinically), was used as a control. ATP release was detected by a bioluminescent kit while HMGB1 and HSP90 relocalization and secretion was detected by fluorescence microscopy and western blotting. VEGF121/rGel-PCI was further investigated as an αCTLA enhancer in CT26 and MC38 tumors by measurement of tumor growth delay. CD8+ Dependent efficacy was evaluated in vivo using a CD8+ antibody.
Results: VEGF121/rGel-PCI was shown to induce increased DAMP signaling as compared to PDT and VEGF121/rGel alone and the magnitude was found similar to that induced by Hypericin-PDT. Furthermore, a significant CD8+ dependent enhanced αCTLA-4 treatment effect was observed when VEGF121/rGel-PCI was used as an adjuvant in both tumor models.
Conclusions: VEGF121/rGel-PCI describes a novel concept for ICI enhancement which induces a rapid CD8+ dependent tumor eradication in both CT26 and MC38 tumors. The concept is based on the combination of intracellular ROS generation and vascular targeting using a plant derived toxin and will be developed towards clinical utilization.
Keywords: immune check point inhibitor (ICI); immunogenic cell death (ICD); photochemical internalization (PCI); photodynamic therapy; targeted toxin; vascular endothelial growth factor; vascular targeting.
Copyright © 2023 Longva, Berg and Weyergang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Light-enhanced VEGF121/rGel: A tumor targeted modality with vascular and immune-mediated efficacy.J Control Release. 2018 Oct 28;288:161-172. doi: 10.1016/j.jconrel.2018.09.005. Epub 2018 Sep 11. J Control Release. 2018. PMID: 30217739
-
Photochemical internalization augments tumor vascular cytotoxicity and specificity of VEGF(121)/rGel fusion toxin.J Control Release. 2014 Apr 28;180:1-9. doi: 10.1016/j.jconrel.2014.02.003. Epub 2014 Feb 13. J Control Release. 2014. PMID: 24531010
-
Corrigendum: Light-enhanced VEGF121/rGel induce immunogenic cell death and increase the antitumor activity of αCTLA4 treatment.Front Immunol. 2024 Jan 9;14:1359973. doi: 10.3389/fimmu.2023.1359973. eCollection 2023. Front Immunol. 2024. PMID: 38264651 Free PMC article.
-
Vascular endothelial growth factor A isoform 121-gelonin fusion protein–conjugated manganese ferrite nanoparticles.2011 Jul 6 [updated 2011 Aug 17]. In: Molecular Imaging and Contrast Agent Database (MICAD) [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2004–2013. 2011 Jul 6 [updated 2011 Aug 17]. In: Molecular Imaging and Contrast Agent Database (MICAD) [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2004–2013. PMID: 21866611 Free Books & Documents. Review.
-
64Cu-Labeled 1,4,7,10-Tetraazacyclododedane-N,N’,N’’,N’’’-tetraacetic acid–conjugated vascular endothelial growth factor A isoform 121-gelonin fusion protein.2011 Jul 6 [updated 2011 Aug 10]. In: Molecular Imaging and Contrast Agent Database (MICAD) [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2004–2013. 2011 Jul 6 [updated 2011 Aug 10]. In: Molecular Imaging and Contrast Agent Database (MICAD) [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2004–2013. PMID: 21850782 Free Books & Documents. Review.
Cited by
-
Cellular Imaging and Time-Domain FLIM Studies of Meso-Tetraphenylporphine Disulfonate as a Photosensitising Agent in 2D and 3D Models.Int J Mol Sci. 2024 Apr 11;25(8):4222. doi: 10.3390/ijms25084222. Int J Mol Sci. 2024. PMID: 38673807 Free PMC article.
-
Photosensitive Hybrid γδ-T Exosomes for Targeted Cancer Photoimmunotherapy.ACS Nano. 2025 Feb 4;19(4):4251-4268. doi: 10.1021/acsnano.4c11024. Epub 2025 Jan 25. ACS Nano. 2025. PMID: 39862206 Free PMC article.
-
Enhancing antitumour immunity with photodynamic therapy.Photochem Photobiol Sci. 2025 Feb;24(2):227-234. doi: 10.1007/s43630-025-00690-2. Epub 2025 Feb 19. Photochem Photobiol Sci. 2025. PMID: 39971873
References
-
- Tarhini AA, Lee SJ, Hodi FS, Rao UNM, Cohen GI, Hamid O, et al. . Phase III study of adjuvant ipilimumab (3 or 10 mg/kg) versus high-dose interferon alfa-2b for resected high-risk melanoma: North american intergroup E1609. J Clin Oncol (2020) 38:567–75. doi: 10.1200/jco.19.01381 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

