Statistical analysis of the storage time of finished product infusion
- PMID: 38180889
- PMCID: PMC10771069
- DOI: 10.1177/03000605231222231
Statistical analysis of the storage time of finished product infusion
Abstract
Objective: This retrospective study determined the storage time of finished infusion in each hospital ward and assessed whether the storage time of finished infusion was within an acceptable range.
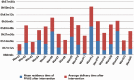
Methods: The research object was the finished infusion (one bag of infusion with only one drug) that is centrally dosed at the Pharmacy Intravenous Admixture Service (PIVAS) of Jiading District Central Hospital Affiliated Shanghai University of Medicine & Health Sciences. We used an automatic scanner to assess the placement time of finished infusion products in various wards of the hospital. We classified the drugs used in various wards, analyzed whether their placement times were reasonable, assessed the reasons for unreasonable placement times, and took intervention measures. Similarly, the storage time of finished infusion was deemed reasonable or unreasonable, the reasons for unreasonable storage times were analyzed, and intervention measures were taken.
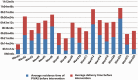
Results: In September 2021, the proportion of infusions stored for an unreasonable time was 12.69%, a decrease of 5.37% compared with August 2021, indicating the effectiveness of intervention measures.
Conclusion: By using statistical analysis and intervention measures, our PIVAS improved the standardized use of finished infusion products and ensured the safety of medication for patients.
Keywords: Pharmacy Intravenous Admixture Service; finished infusion; hospital ward; intravenous medicine; placement time; quality; storage time.
Conflict of interest statement
Declaration of conflicting interestsThe authors declare that they have no conflict of interest.
Figures
Similar articles
-
Application of information-intelligence technologies in pharmacy intravenous admixture services in a Chinese third-class a hospital.BMC Health Serv Res. 2022 Oct 7;22(1):1238. doi: 10.1186/s12913-022-08580-4. BMC Health Serv Res. 2022. PMID: 36207708 Free PMC article.
-
Real-world study of the leakage of two types of infusion bags in multicenter pharmacy intravenous admixture service (PIVAS).Front Pharmacol. 2023 Oct 6;14:1273020. doi: 10.3389/fphar.2023.1273020. eCollection 2023. Front Pharmacol. 2023. PMID: 37869755 Free PMC article.
-
Development of regional pharmacy intravenous admixture services data reporting and analysis platform for enhanced quality control ability.BMC Health Serv Res. 2024 Feb 22;24(1):231. doi: 10.1186/s12913-024-10696-8. BMC Health Serv Res. 2024. PMID: 38388389 Free PMC article.
-
Construction situation, costs and charges associated with pharmacy intravenous admixture services: multi-center cross-sectional survey based on 137 medical institutions in mainland China.BMC Health Serv Res. 2020 Jun 24;20(1):577. doi: 10.1186/s12913-020-05336-w. BMC Health Serv Res. 2020. PMID: 32580775 Free PMC article.
-
Status analysis of quality control of administered infusion solution with cytotoxic drugs.Oncol Rev. 2025 Jan 6;18:1415677. doi: 10.3389/or.2024.1415677. eCollection 2024. Oncol Rev. 2025. PMID: 39834531 Free PMC article. Review.
References
-
- Yang CS, Lin YZ, Zhang LL, et al.. Evidence-based evaluation of construction status of pharmacy intravenous admixture services in China [J]. Journal of Pediatric Pharmacy 2020; 26: 32–35.
-
- Huang J, Wu WY, Jiang ZF, et al.. Application of drug safety management system based on closed-loop concept [J]. Nursing Safety 2018; 18: 1103–1107.
-
- Yuan ZZ, Li QQ, Tang TT, et al.. Studies on the optimization of decontamination protocol for surfaces contaminated with cytotoxic drugs in PIVAS [J]. J Oncol Pharm Pract 2022; 12. - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

