Protein tyrosine phosphatase PTPRO represses lung adenocarcinoma progression by inducing mitochondria-dependent apoptosis and restraining tumor metastasis
- PMID: 38182570
- PMCID: PMC10770368
- DOI: 10.1038/s41419-023-06375-x
Protein tyrosine phosphatase PTPRO represses lung adenocarcinoma progression by inducing mitochondria-dependent apoptosis and restraining tumor metastasis
Abstract
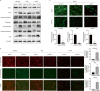
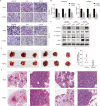
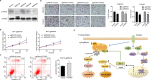
Emerging evidence indicates that protein activities regulated by receptor protein tyrosine phosphatases (RPTPs) are crucial for a variety of cellular processes, such as proliferation, apoptosis, and immunological response. Protein tyrosine phosphatase receptor type O (PTPRO), an RPTP, has been revealed as a putative suppressor in the development of particular tumors. However, the function and the underlying mechanisms of PTPRO in regulating of lung adenocarcinoma (LUAD) are not well understood. In this view, the present work investigated the role of PTPRO in LUAD. Analysis of 90 pairs of clinical LUAD specimens revealed significantly lower PTPRO levels in LUAD compared with adjacent non-tumor tissue, as well as a negative correlation of PTPRO expression with tumor size and TNM stage. Survival analyses demonstrated that PTPRO level can help stratify the prognosis of LUAD patients. Furthermore, PTPRO overexpression was found to suppress the progression of LUAD both in vitro and in vivo by inducing cell death via mitochondria-dependent apoptosis, downregulating protein expression of molecules (Bcl-2, Bax, caspase 3, cleaved-caspase 3/9, cleaved-PARP and Bid) essential in cell survival. Additionally, PTPRO decreased LUAD migration and invasion by regulating proteins involved in the epithelial-to-mesenchymal transition (E-cadherin, N-cadherin, and Snail). Moreover, PTPRO was shown to restrain JAK2/STAT3 signaling pathways. Expression of PTPRO was negatively correlated with p-JAK2, p-STAT3, Bcl-2, and Snail levels in LUAD tumor samples. Furthermore, the anti-tumor effect of PTPRO in LUAD was significant but compromised in STAT3-deficient cells. These data support the remarkable suppressive role of PTPRO in LUAD, which may represent a viable therapeutic target for LUAD patients.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures





Similar articles
-
Comprehensively Analyze the Prognosis Significance and Immune Implication of PTPRO in Lung Adenocarcinoma.Mediators Inflamm. 2023 Feb 9;2023:5248897. doi: 10.1155/2023/5248897. eCollection 2023. Mediators Inflamm. 2023. PMID: 36816740 Free PMC article.
-
IL-6 promotes PD-L1 expression in monocytes and macrophages by decreasing protein tyrosine phosphatase receptor type O expression in human hepatocellular carcinoma.J Immunother Cancer. 2020 Jun;8(1):e000285. doi: 10.1136/jitc-2019-000285. J Immunother Cancer. 2020. PMID: 32581055 Free PMC article.
-
Estrogen-sensitive PTPRO expression represses hepatocellular carcinoma progression by control of STAT3.Hepatology. 2013 Feb;57(2):678-88. doi: 10.1002/hep.25980. Epub 2012 Oct 30. Hepatology. 2013. PMID: 22821478
-
Tumor-Suppression Mechanisms of Protein Tyrosine Phosphatase O and Clinical Applications.Asian Pac J Cancer Prev. 2015;16(15):6215-23. doi: 10.7314/apjcp.2015.16.15.6215. Asian Pac J Cancer Prev. 2015. PMID: 26434819 Review.
-
Regulatory Functions of Protein Tyrosine Phosphatase Receptor Type O in Immune Cells.Front Immunol. 2021 Nov 22;12:783370. doi: 10.3389/fimmu.2021.783370. eCollection 2021. Front Immunol. 2021. PMID: 34880876 Free PMC article. Review.
Cited by
-
PTPRO represses breast cancer lung metastasis by inhibiting the JAK2-YAP axis.Sci Rep. 2025 Feb 27;15(1):7065. doi: 10.1038/s41598-025-91341-0. Sci Rep. 2025. PMID: 40016288 Free PMC article.
-
ADRB2 is regulated by TRIM22 and facilitates lung adenocarcinoma progression via JAK2/STAT3 signaling pathway.Sci Rep. 2025 Jul 1;15(1):22083. doi: 10.1038/s41598-025-06017-6. Sci Rep. 2025. PMID: 40594822 Free PMC article.
-
Paracetamol Induces Apoptosis, Reduces Colony Formation, and Increases PTPRO Gene Expression in Human Embryonic Kidney HEK 293 Cells.J Biochem Mol Toxicol. 2025 Aug;39(8):e70366. doi: 10.1002/jbt.70366. J Biochem Mol Toxicol. 2025. PMID: 40787714 Free PMC article.
References
-
- Thomas PE, Wharram BL, Goyal M, Wiggins JE, Holzman LB, Wiggins RC. GLEPP1, a renal glomerular epithelial cell (podocyte) membrane protein-tyrosine phosphatase. Identification, molecular cloning, and characterization in rabbit. J Biol Chem. 1994;269:19953–62. doi: 10.1016/S0021-9258(17)32113-0. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

