Impact of tissue penetration and albumin binding on design of T cell targeted bispecific agents
- PMID: 38183712
- PMCID: PMC10809211
- DOI: 10.1016/j.neo.2023.100962
Impact of tissue penetration and albumin binding on design of T cell targeted bispecific agents
Abstract
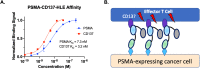
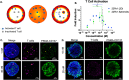
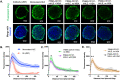
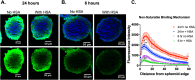
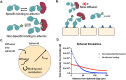
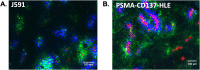
Bispecific agents are a rapidly growing class of cancer therapeutics, and immune targeted bispecific agents have the potential to expand functionality well beyond monoclonal antibody agents. Humabodies⁎ are fully human single domain antibodies that can be linked in a modular fashion to form multispecific therapeutics. However, the effect of heterogeneous delivery on the efficacy of crosslinking bispecific agents is currently unclear. In this work, we utilize a PSMA-CD137 Humabody with an albumin binding half-life extension (HLE) domain to determine the impact of tissue penetration on T cell activating bispecific agents. Using heterotypic spheroids, we demonstrate that increased tissue penetration results in higher T cell activation at sub-saturating concentrations. Next, we tested the effect of two different albumin binding moieties on tissue distribution using albumin-specific HLE domains with varying affinities for albumin and a non-specific lipophilic dye. The results show that a specific binding mechanism to albumin does not influence tissue penetration, but a non-specific mechanism reduced both spheroid uptake and distribution in the presence of albumin. These results highlight the potential importance of tissue penetration on bispecific agent efficacy and describe how the design parameters including albumin-binding domains can be selected to maximize the efficacy of bispecific agents.
Keywords: Bispecific antibodies; Heterotypic spheroids; Single domain antibodies; T-cell activating agents; Tumoral distribution.
Copyright © 2023. Published by Elsevier Inc.
Conflict of interest statement
Declaration of competing interest The authors declare the following financial interests/personal relationships which may be considered as potential competing interests: Colette Johnston, Steven Vance, James Legg, and Laurie Galson-Holt are employees of Crescendo Biologics, which is developing CB307 and related therapeutics. All other authors declare no competing interests.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

