Regional disparities in the availability of cancer clinical trials in Korea
- PMID: 38186251
- PMCID: PMC11040215
- DOI: 10.4178/epih.e2024006
Regional disparities in the availability of cancer clinical trials in Korea
Abstract
Objectives: Unequal access to cancer clinical trials is an important issue, given the potential benefits of participation for cancer patients. We evaluated regional disparities in access to cancer clinical trials in Korea.
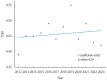
Methods: From the Ministry of Food and Drug Safety database, we extracted 2,465 records of all cancer clinical trials approved between January 2012 and April 2023. To measure disparities in cancer clinical trial access, we calculated the ratio of clinical trials open to non-capital areas relative to those open to capital areas. We then analyzed temporal trends in this ratio, which we termed the trial geographical equity index (TGEI).
Results: Disparities in access to cancer clinical trials, as indicated by the TGEI, did not significantly improve during the study period (regression coefficient, 0.002; p=0.59). However, for phase II/III trials sponsored by global pharmaceutical companies, the TGEI improved significantly (regression coefficient, 0.021; p<0.01). In contrast, the TGEI deteriorated for trials initiated by investigators or those testing domestically developed therapeutics (regression coefficient, -0.015; p=0.05). Furthermore, the increasing trend of TGEI for phase II/III trials sponsored by global companies began to reverse after 2019, coinciding with the outbreak of coronavirus disease 2019 (COVID-19).
Conclusions: Over the past decade, access to cancer clinical trials has improved in Korea, particularly for phase II/III trials evaluating therapeutics from global companies. However, this increase in accessibility has not extended to trials initiated by investigators or those assessing domestically developed therapeutics. Additionally, the impact of COVID-19 on disparities in clinical trial access should be closely monitored.
Keywords: Clinical trial; Health inequities; Health services accessibility; Healthcare disparities; Neoplasms.
Conflict of interest statement
The authors have no conflicts of interest to declare for this study.
Figures


Similar articles
-
The Impact of COVID-19 on the Conduct of Clinical Trials for Medical Products in Korea.J Korean Med Sci. 2020 Sep 14;35(36):e329. doi: 10.3346/jkms.2020.35.e329. J Korean Med Sci. 2020. PMID: 32924344 Free PMC article.
-
Trends and disparities in avoidable, treatable, and preventable mortalities in South Korea, 2001-2020: comparison of capital and non-capital areas.Epidemiol Health. 2022;44:e2022067. doi: 10.4178/epih.e2022067. Epub 2022 Aug 16. Epidemiol Health. 2022. PMID: 35989656 Free PMC article.
-
Regional disparities in major cancer incidence in Korea, 1999-2018.Epidemiol Health. 2023;45:e2023089. doi: 10.4178/epih.e2023089. Epub 2023 Oct 12. Epidemiol Health. 2023. PMID: 37857340 Free PMC article.
-
Efaproxiral: GSJ 61, JP 4, KDD 86, RS 4, RSR 13.Drugs R D. 2005;6(3):178-85. doi: 10.2165/00126839-200506030-00007. Drugs R D. 2005. PMID: 15869322 Review.
-
INGN 201: Ad-p53, Ad5CMV-p53, adenoviral p53, p53 gene therapy--introgen, RPR/INGN 201.Drugs R D. 2007;8(3):176-87. doi: 10.2165/00126839-200708030-00005. Drugs R D. 2007. PMID: 17472413 Review.
Cited by
-
Impact of fragmented care on cancer outcomes among korean women with breast and cervical cancer: a focus on regional and economic disparities.Int J Equity Health. 2025 Jul 3;24(1):192. doi: 10.1186/s12939-025-02542-y. Int J Equity Health. 2025. PMID: 40611114 Free PMC article.
References
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

