This is a preprint.
Branched-chain keto acids promote an immune-suppressive and neurodegenerative microenvironment in leptomeningeal disease
- PMID: 38187773
- PMCID: PMC10769272
- DOI: 10.1101/2023.12.18.572239
Branched-chain keto acids promote an immune-suppressive and neurodegenerative microenvironment in leptomeningeal disease
Abstract
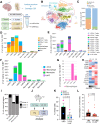
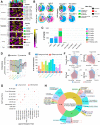
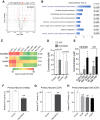
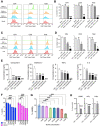
Leptomeningeal disease (LMD) occurs when tumors seed into the leptomeningeal space and cerebrospinal fluid (CSF), leading to severe neurological deterioration and poor survival outcomes. We utilized comprehensive multi-omics analyses of CSF from patients with lymphoma LMD to demonstrate an immunosuppressive cellular microenvironment and identified dysregulations in proteins and lipids indicating neurodegenerative processes. Strikingly, we found a significant accumulation of toxic branched-chain keto acids (BCKA) in the CSF of patients with LMD. The BCKA accumulation was found to be a pan-cancer occurrence, evident in lymphoma, breast cancer, and melanoma LMD patients. Functionally, BCKA disrupted the viability and function of endogenous T lymphocytes, chimeric antigen receptor (CAR) T cells, neurons, and meningeal cells. Treatment of LMD mice with BCKA-reducing sodium phenylbutyrate significantly improved neurological function, survival outcomes, and efficacy of anti-CD19 CAR T cell therapy. This is the first report of BCKA accumulation in LMD and provides preclinical evidence that targeting these toxic metabolites improves outcomes.
Keywords: Brain; Branched-chain keto acids; CSF; Leptomeninges.
Conflict of interest statement
IS, MLK, YR, RK, OO, HA, TR, BE, GCW, ZC, YAC, ML, LNFD, SAC, PAS, and VI declare no relevant conflicts of interest. MDJ would like to disclose consultancy for Kite/Gilead and Novartis and research funding from Kite/Gilead, Incyte, and Loxo@Lilly. PAF would like to disclose consultancy with AbbVie Inc., Bristol-Myers Squibb, Boehringer-Ingelheim, NCI Neuro-Oncology Branch Peer Review, NCRI, NIH, Novellus, Physical Sciences Oncology Network, Tocagen (not active), Ziopharm, National Brain Tumor Society. He is also on the advisory board for Bayer, BTG, GlaxoSmithKline (GSK), Inovio, Novocure, AnHeart Therapeutics, and Midatech. MH would like to disclose that Moffitt Cancer Center has licensed Intellectual Property (IP) related to the proliferation and expansion of tumor-infiltrating lymphocytes (TILs) to Iovance Biotherapeutics. MH is a co-inventor on such Intellectual Property. MH reports common stock holdings in AbbVie, Inc., Amgen, Inc., BioHaven Pharmaceuticals, and Bristol Myers Squibb. SPT would like to disclose that Moffitt Cancer Center has licensed Intellectual Property (IP) related to the proliferation and expansion of tumor-infiltrating lymphocytes (TILs) to Iovance Biotherapeutics. Moffitt has also licensed IP to Tuhura Biopharma. Dr. Pilon-Thomas (SPT) is an inventor on such Intellectual Property. SPT is listed as a co-inventor on a patent application with Provectus Biopharmaceuticals. SPT participates in sponsored research agreements with Provectus Biopharmaceuticals, Iovance Biotherapeutics, Intellia Therapeutics, Dyve Biosciences, Turnstone Biologics, and Celgene that are not related to this research. SPT has received research support that is not related to this research from the following entities: NIHNCI, DOD, Swim Across America, the V Foundation, and The Mark Foundation for Cancer Research. SPT has received consulting fees from Seagen Inc., Morphogenesis, Inc., and KSQ Therapeutics. FLL would like to disclose the following: Scientific Advisory Role/Consulting Fees: A2, Allogene, Amgen, Bluebird Bio, BMS, Calibr, Caribou, Cowen, EcoR1, Gerson Lehrman Group (GLG), Iovance, Kite Pharma, Janssen, Legend Biotech, Novartis, Sana, Umoja, Pfizer. Data Safety Monitoring Board: Data and Safety Monitoring Board for the NCI Safety Oversight CAR T-cell Therapies Committee. Research Contracts or Grants to my Institution for Service: Kite Pharma (Institutional), Allogene (Institutional), CERo Therapeutics (Institutional), Novartis (Institutional), BlueBird Bio (Institutional), 2SeventyBio (Institutional), BMS (Institutional), National Cancer Institute (R01CA244328 MPI: Locke; P30CA076292 PI: Cleveland), Leukemia and Lymphoma Society Scholar in Clinical Research (PI: Locke). Patents, Royalties, Other Intellectual Property: Several patents held by the institution in my name (unlicensed) in the field of cellular immunotherapy. Education or Editorial Activity: Aptitude Health, ASH, BioPharma Communications CARE Education, Clinical Care Options Oncology, Imedex, Society for Immunotherapy of Cancer
Figures

Similar articles
-
Leptomeningeal Disease (LMD) in Patients with Melanoma Metastases.Cancers (Basel). 2023 Mar 21;15(6):1884. doi: 10.3390/cancers15061884. Cancers (Basel). 2023. PMID: 36980770 Free PMC article. Review.
-
A preclinical model of patient-derived cerebrospinal fluid circulating tumor cells for experimental therapeutics in leptomeningeal disease from melanoma.Neuro Oncol. 2022 Oct 3;24(10):1673-1686. doi: 10.1093/neuonc/noac054. Neuro Oncol. 2022. PMID: 35213727 Free PMC article.
-
Leptomeningeal Metastases in Melanoma Patients: An Update on and Future Perspectives for Diagnosis and Treatment.Int J Mol Sci. 2023 Jul 14;24(14):11443. doi: 10.3390/ijms241411443. Int J Mol Sci. 2023. PMID: 37511202 Free PMC article. Review.
-
Leptomeningeal Metastasis: A Review of the Pathophysiology, Diagnostic Methodology, and Therapeutic Landscape.Curr Oncol. 2023 Jun 19;30(6):5906-5931. doi: 10.3390/curroncol30060442. Curr Oncol. 2023. PMID: 37366925 Free PMC article. Review.
-
Metabolic flux analysis of branched-chain amino and keto acids (BCAA, BCKA) and β-hydroxy β-methylbutyric acid across multiple organs in the pig.Am J Physiol Endocrinol Metab. 2021 Mar 1;320(3):E629-E640. doi: 10.1152/ajpendo.00384.2020. Epub 2021 Feb 1. Am J Physiol Endocrinol Metab. 2021. PMID: 33522397
Cited by
-
Leptomeningeal metastatic disease: new frontiers and future directions.Nat Rev Clin Oncol. 2025 Feb;22(2):134-154. doi: 10.1038/s41571-024-00970-3. Epub 2024 Dec 9. Nat Rev Clin Oncol. 2025. PMID: 39653782 Review.
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
