Exploring individual fixel-based white matter abnormalities in epilepsy
- PMID: 38187877
- PMCID: PMC10768884
- DOI: 10.1093/braincomms/fcad352
Exploring individual fixel-based white matter abnormalities in epilepsy
Abstract
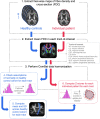
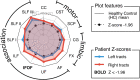
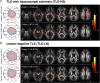
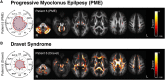
Diffusion MRI has provided insight into the widespread structural connectivity changes that characterize epilepsies. Although syndrome-specific white matter abnormalities have been demonstrated, studies to date have predominantly relied on statistical comparisons between patient and control groups. For diffusion MRI techniques to be of clinical value, they should be able to detect white matter microstructural changes in individual patients. In this study, we apply an individualized approach to a technique known as fixel-based analysis, to examine fibre-tract-specific abnormalities in individuals with epilepsy. We explore the potential clinical value of this individualized fixel-based approach in epilepsy patients with differing syndromic diagnoses. Diffusion MRI data from 90 neurologically healthy control participants and 10 patients with epilepsy (temporal lobe epilepsy, progressive myoclonus epilepsy, and Dravet Syndrome, malformations of cortical development) were included in this study. Measures of fibre density and cross-section were extracted for all participants across brain white matter fixels, and mean values were computed within select tracts-of-interest. Scanner harmonized and normalized data were then used to compute Z-scores for individual patients with epilepsy. White matter abnormalities were observed in distinct patterns in individual patients with epilepsy, both at the tract and fixel level. For patients with specific epilepsy syndromes, the detected white matter abnormalities were in line with expected syndrome-specific clinical phenotypes. In patients with lesional epilepsies (e.g. hippocampal sclerosis, periventricular nodular heterotopia, and bottom-of-sulcus dysplasia), white matter abnormalities were spatially concordant with lesion location. This proof-of-principle study demonstrates the clinical potential of translating advanced diffusion MRI methodology to individual-patient-level use in epilepsy. This technique could be useful both in aiding diagnosis of specific epilepsy syndromes, and in localizing structural abnormalities, and is readily amenable to other neurological disorders. We have included code and data for this study so that individualized white matter changes can be explored robustly in larger cohorts in future work.
Keywords: diffusion MRI; epilepsy; fixel-based analysis; individual analysis; white matter.
© The Author(s) 2023. Published by Oxford University Press on behalf of the Guarantors of Brain.
Conflict of interest statement
The authors have no competing interests to declare.
Figures

Similar articles
-
White matter abnormalities across different epilepsy syndromes in adults: an ENIGMA-Epilepsy study.Brain. 2020 Aug 1;143(8):2454-2473. doi: 10.1093/brain/awaa200. Brain. 2020. PMID: 32814957 Free PMC article.
-
White matter abnormalities characterize the acute stage of sports-related mild traumatic brain injury.Brain Commun. 2022 Aug 17;4(4):fcac208. doi: 10.1093/braincomms/fcac208. eCollection 2022. Brain Commun. 2022. PMID: 36043140 Free PMC article.
-
Longitudinal fibre-specific white matter damage predicts cognitive decline in multiple sclerosis.Brain Commun. 2024 Jan 27;6(1):fcae018. doi: 10.1093/braincomms/fcae018. eCollection 2024. Brain Commun. 2024. PMID: 38344654 Free PMC article.
-
Fixel-based Analysis of Diffusion MRI: Methods, Applications, Challenges and Opportunities.Neuroimage. 2021 Nov 1;241:118417. doi: 10.1016/j.neuroimage.2021.118417. Epub 2021 Jul 21. Neuroimage. 2021. PMID: 34298083 Review.
-
Histological and MRI markers of white matter damage in focal epilepsy.Epilepsy Res. 2018 Feb;140:29-38. doi: 10.1016/j.eplepsyres.2017.11.010. Epub 2017 Nov 23. Epilepsy Res. 2018. PMID: 29227798 Review.
Cited by
-
Individual-level analysis of MRI T2 relaxometry in mild traumatic brain injury: Possible indications of brain inflammation.Neuroimage Clin. 2024;43:103647. doi: 10.1016/j.nicl.2024.103647. Epub 2024 Jul 22. Neuroimage Clin. 2024. PMID: 39068788 Free PMC article.
-
Microangiopathy in temporal lobe epilepsy with diffusion MRI alterations and cognitive decline.Acta Neuropathol. 2024 Oct 8;148(1):49. doi: 10.1007/s00401-024-02809-8. Acta Neuropathol. 2024. PMID: 39377933 Free PMC article.
-
Juvenile Myoclonic Epilepsy Imaging Endophenotypes and Relationship With Cognition and Resting-State EEG.Hum Brain Mapp. 2025 May;46(7):e70226. doi: 10.1002/hbm.70226. Hum Brain Mapp. 2025. PMID: 40347042 Free PMC article.
-
Identification of LAMA2 compound heterozygous variants: a case report.Transl Pediatr. 2024 Jun 30;13(6):1001-1006. doi: 10.21037/tp-24-62. Epub 2024 Jun 14. Transl Pediatr. 2024. PMID: 38984033 Free PMC article.
-
Diffusion-Weighted Magnetic Resonance Imaging: A Diagnostic Tool for Auditory (Axonal) Neuropathy.Eur J Neurol. 2025 Feb;32(2):e70083. doi: 10.1111/ene.70083. Eur J Neurol. 2025. PMID: 39932015 Free PMC article.
References
-
- Campos BM, Coan AC, Beltramini GC, et al. White matter abnormalities associate with type and localization of focal epileptogenic lesions. Epilepsia. 2015;56(1):125–132. - PubMed
-
- Vaughan DN, Raffelt D, Curwood E, et al. Tract-specific atrophy in focal epilepsy: Disease, genetics, or seizures? Ann Neurol. 2017;81(2):240–250. - PubMed
LinkOut - more resources
Full Text Sources
