New hope for esophageal stricture prevention: A prospective single-center trial on acellular dermal matrix
- PMID: 38187918
- PMCID: PMC10768038
- DOI: 10.4253/wjge.v15.i12.725
New hope for esophageal stricture prevention: A prospective single-center trial on acellular dermal matrix
Abstract
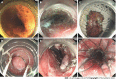
Background: Given the high incidence of esophageal cancer in China, an increasing number of patients there are undergoing endoscopic mucosal dissection (ESD). Although the 5-year survival rate after ESD can exceed 95%, esophageal stricture, the most common and serious postoperative complication, affects the long-term prognosis of patients and the quality of life. Autologous mucosal grafts have proven to be successful in preventing stricture after ESD for early esophageal cancer.
Aim: To examine the viability of acellular dermal matrix (ADM) as an alternative to autologous mucosa for the prevention of stricture after ESD.
Methods: This is a prospective, single-center, controlled study. Consecutive patients who underwent ESD surgery and were willing to undergo autologous mucosal transplantation were recruited between January 1 and December 31, 2017. Consecutive patients who underwent ESD surgery and were willing to undergo ADM transplantation were recruited between January 1 to December 31, 2019. A final three-year follow-up of patients who received transplants was conducted.
Results: Based on the current incidence of esophageal stricture, the sample size required for both the autologous mucosal graft group and the ADM group was calculated to be 160 cases. Due to various factors, a total of 20 patients with autologous mucosal grafts and 25 with ADM grafts were recruited. Based on the inclusion exclusion and withdrawal criteria, 9 patients ultimately received autologous mucosal grafts and completed the follow-up, while 11 patients received ADM grafts and completed the follow-up. Finally, there were 2 cases of stenosis in the autologous mucosal transplantation group with a stenosis rate of 22.22% and 2 cases of stenosis in the ADM transplantation group with a stenosis rate of 18.18%, with no significant difference noted between the groups (P = 0.94).
Conclusion: In this prospective, single-center, controlled trial, we compared the effectiveness of autologous mucosa transplantation and ADM for the prevention of esophageal stricture. Due to certain condition limitations, we were unable to recruit sufficient subjects meeting our target requirements. However, we implemented strict inclusion, exclusion, and withdrawal criteria and successfully completed three years of follow-up, resulting in valuable clinical insights. Based on our findings, we hypothesize that ADM may be similarly effective to autologous mucosal transplantation in the prevention of esophageal stricture, offering a comparable and alternative approach. This study provides a new therapeutic idea and direction for the prevention of esophageal stricture.
Keywords: Duodenal subepithelial lesion; Endoscopic resection; Over-the-scope clip; Perforation.
©The Author(s) 2023. Published by Baishideng Publishing Group Inc. All rights reserved.
Conflict of interest statement
Conflict-of-interest statement: All the authors declare that they have no conflict interests to disclose.
Figures
Similar articles
-
Esophageal mucosal auto-transplantation versus acellular dermal matrix transplantation for preventing esophageal stenosis after endoscopic resection in patients with superficial esophageal mucosal lesions.Surg Endosc. 2025 Jun;39(6):3760-3768. doi: 10.1007/s00464-025-11718-0. Epub 2025 May 5. Surg Endosc. 2025. PMID: 40325245
-
Acellular dermal matrix for esophageal stricture prevention after endoscopic submucosal dissection in a porcine model.Gastrointest Endosc. 2017 Dec;86(6):1160-1167. doi: 10.1016/j.gie.2017.02.038. Epub 2017 Apr 8. Gastrointest Endosc. 2017. PMID: 28396274
-
Transplantation of acellularized dermis matrix (ADM) plus fully covered metal stent to prevent stricture after circumferential endoscopic submucosal dissection of early esophageal cancer (with video).Regen Ther. 2021 Oct 23;18:441-446. doi: 10.1016/j.reth.2021.10.002. eCollection 2021 Dec. Regen Ther. 2021. PMID: 34754889 Free PMC article.
-
Prevention of Stricture after Endoscopic Submucosal Dissection for Superficial Esophageal Cancer: A Review of the Literature.J Clin Med. 2020 Dec 23;10(1):20. doi: 10.3390/jcm10010020. J Clin Med. 2020. PMID: 33374780 Free PMC article. Review.
-
Esophageal Stricture Prevention after Endoscopic Submucosal Dissection.Clin Endosc. 2016 May;49(3):241-56. doi: 10.5946/ce.2015.099. Epub 2016 Mar 7. Clin Endosc. 2016. PMID: 26949124 Free PMC article. Review.
Cited by
-
[Preparation and in vivo osteogenesis of acellular dermal matrix/dicalcium phosphate composite scaffold for bone repair].Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2024 Jun 15;38(6):755-762. doi: 10.7507/1002-1892.202403059. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2024. PMID: 38918199 Free PMC article. Chinese.
References
-
- Bourke MJ, Neuhaus H, Bergman JJ. Endoscopic Submucosal Dissection: Indications and Application in Western Endoscopy Practice. Gastroenterology. 2018;154:1887–1900.e5. - PubMed
-
- Isomoto H, Yamaguchi N, Nakayama T, Hayashi T, Nishiyama H, Ohnita K, Takeshima F, Shikuwa S, Kohno S, Nakao K. Management of esophageal stricture after complete circular endoscopic submucosal dissection for superficial esophageal squamous cell carcinoma. BMC Gastroenterol. 2011;11:46. - PMC - PubMed
-
- Miwata T, Oka S, Tanaka S, Kagemoto K, Sanomura Y, Urabe Y, Hiyama T, Chayama K. Risk factors for esophageal stenosis after entire circumferential endoscopic submucosal dissection for superficial esophageal squamous cell carcinoma. Surg Endosc. 2016;30:4049–4056. - PubMed
-
- Oliveira JF, Moura EG, Bernardo WM, Ide E, Cheng S, Sulbaran M, Santos CM, Sakai P. Prevention of esophageal stricture after endoscopic submucosal dissection: a systematic review and meta-analysis. Surg Endosc. 2016;30:2779–2791. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

