Postsynaptic dorsal column pathway activation during spinal cord stimulation in patients with chronic pain
- PMID: 38188030
- PMCID: PMC10771283
- DOI: 10.3389/fnins.2023.1297814
Postsynaptic dorsal column pathway activation during spinal cord stimulation in patients with chronic pain
Abstract
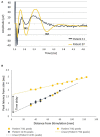
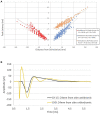
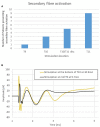
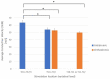
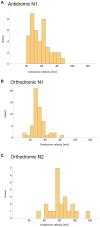
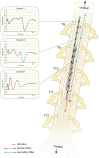
Spinal cord stimulation (SCS) treatment for chronic pain relies on the activation of primary sensory fibres ascending to the brain in the dorsal columns. While the efficacy of SCS has been demonstrated, the precise mechanism of action and nature of the fibres activated by stimulation remain largely unexplored. Our investigation in humans with chronic neuropathic pain undergoing SCS therapy, found that post-synaptic dorsal column (PSDC) fibres can be activated synaptically by the primary afferents recruited by stimulation, and axonically by the stimulation pulses directly. Synaptic activation occurred in 9 of the 14 patients analysed and depended on the vertebral level of stimulation. A clear difference in conduction velocities between the primary afferents and the PSDC fibres were observed. Identification of PSDC fibre activation in humans emphasises the need for further investigation into the role they play in pain relief and the sensory response sensation (paraesthesia) experienced by patients undergoing SCS.
Keywords: electrophysiology; mechanism of action; neural response recordings; neuroanatomy; neuropathic pain; postsynaptic dorsal column pathway; spinal cord stimulation.
Copyright © 2023 Gmel, Santos Escapa, Benkohen, Mugan, Parker and Palmisani.
Conflict of interest statement
GG, RS, TB, DM, and JP were employees of Saluda Medical at the time of the study. GG is the inventor of a patent assigned to Saluda Medical related to the material in this publication (WO2021146778A1). SP has received honoraria for speaker engagements and participated in scientific advisory boards for Saluda Medical and Nevro.
Figures

References
LinkOut - more resources
Full Text Sources

