Zolbetuximab for Claudin18.2-positive gastric or gastroesophageal junction cancer
- PMID: 38188462
- PMCID: PMC10768589
- DOI: 10.1177/17588359231217967
Zolbetuximab for Claudin18.2-positive gastric or gastroesophageal junction cancer
Abstract
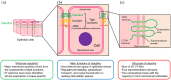
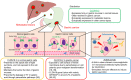
Claudins (CLDNs) are a family of major membrane proteins that form components of tight junctions. In normal tissues, CLDNs seal the intercellular space in the epithelial sheets to regulate tissue permeability, paracellular transport, and signal transduction. Claudin18.2 (CLDN18.2), a member of the CLDN family, is expressed specifically in gastric mucosal cells in normal tissue, and its expression is often retained in gastric cancer cells. CLDN18.2 is ectopically expressed in many cancers other than gastric cancer such as esophageal cancer, pancreatic cancer, biliary tract cancer, non-small-cell lung cancer, and ovarian cancer. Structurally, CLDN18.2 is localized on the apical side of the cell membrane and has extracellular loops capable of binding monoclonal antibodies. Upon malignant transformation, CLDN18.2 is exposed to the cell surface of the whole membrane, which enables the binding of monoclonal antibodies. Based on these characteristics, CLDN18.2 was considered to be optimal for target therapy, and zolbetuximab was developed which is a first-in-class chimeric immunoglobulin G1 monoclonal antibody highly specific for CLDN18.2. It binds to CLDN18.2 on the tumor cell surface and stimulates cellular and soluble immune effectors that activate antibody-dependent cytotoxicity and complement-dependent cytotoxicity. Recently, zolbetuximab combined with chemotherapy demonstrated a survival benefit in patients with CLDN18.2-positive and HER-2-negative gastric or gastroesophageal junction cancers in the global phase III SPOTLIGHT and GLOW trials. From these clinically meaningful results, CLDN18.2-targeting therapy including zolbetuximab has attracted a lot of attention. In this review, we summarize the clinical implications of CLDN18.2-positive gastric or GEJ cancer, and CLDN18.2-targeting therapy, mainly for zolbetuximab.
Keywords: Claudin18.2; Zolbetuximab; gastric cancer; gastroesophageal junction cancer.
© The Author(s), 2024.
Figures
Similar articles
-
Zolbetuximab for Unresectable and Metastatic Gastric and Gastroesophageal Junction Adenocarcinoma: A Review of Literature.Cureus. 2024 Dec 6;16(12):e75206. doi: 10.7759/cureus.75206. eCollection 2024 Dec. Cureus. 2024. PMID: 39759684 Free PMC article. Review.
-
FAST: a randomised phase II study of zolbetuximab (IMAB362) plus EOX versus EOX alone for first-line treatment of advanced CLDN18.2-positive gastric and gastro-oesophageal adenocarcinoma.Ann Oncol. 2021 May;32(5):609-619. doi: 10.1016/j.annonc.2021.02.005. Epub 2021 Feb 19. Ann Oncol. 2021. PMID: 33610734 Clinical Trial.
-
Effect of anti-claudin 18.2 monoclonal antibody zolbetuximab alone or combined with chemotherapy or programmed cell death-1 blockade in syngeneic and xenograft gastric cancer models.J Pharmacol Sci. 2024 Jul;155(3):84-93. doi: 10.1016/j.jphs.2024.04.004. Epub 2024 Apr 18. J Pharmacol Sci. 2024. PMID: 38797537
-
Zolbetuximab plus chemotherapy for locally advanced unresectable or metastatic stomach or gastroesophageal junction cancers: a plain language summary.Future Oncol. 2024;20(26):1861-1877. doi: 10.1080/14796694.2024.2342107. Epub 2024 May 24. Future Oncol. 2024. PMID: 38861294 Free PMC article.
-
Zolbetuximab-clzb: Targeting Claudin 18.2 in Advanced Gastric and Gastroesophageal Adenocarcinoma.Ann Pharmacother. 2025 May 27:10600280251342801. doi: 10.1177/10600280251342801. Online ahead of print. Ann Pharmacother. 2025. PMID: 40426305 Review.
Cited by
-
Protein isoform-centric therapeutics: expanding targets and increasing specificity.Nat Rev Drug Discov. 2024 Oct;23(10):759-779. doi: 10.1038/s41573-024-01025-z. Epub 2024 Sep 4. Nat Rev Drug Discov. 2024. PMID: 39232238 Review.
-
Claudin 18.2: An attractive marker in pancreatic ductal adenocarcinoma.Oncol Lett. 2025 Jan 14;29(3):140. doi: 10.3892/ol.2025.14886. eCollection 2025 Mar. Oncol Lett. 2025. PMID: 39850722 Free PMC article.
-
Discovery of Therapeutic Antibodies Targeting Complex Multi-Spanning Membrane Proteins.BioDrugs. 2024 Nov;38(6):769-794. doi: 10.1007/s40259-024-00682-1. Epub 2024 Oct 25. BioDrugs. 2024. PMID: 39453540 Free PMC article. Review.
-
Practical Management of Zolbetuximab Administration: The Project VYLOY Initiative.Cancers (Basel). 2025 Jun 15;17(12):1996. doi: 10.3390/cancers17121996. Cancers (Basel). 2025. PMID: 40563646 Free PMC article.
-
Zolbetuximab for Unresectable and Metastatic Gastric and Gastroesophageal Junction Adenocarcinoma: A Review of Literature.Cureus. 2024 Dec 6;16(12):e75206. doi: 10.7759/cureus.75206. eCollection 2024 Dec. Cureus. 2024. PMID: 39759684 Free PMC article. Review.
References
-
- Sung H, Ferlay J, Siegel RL, et al.. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2021; 71: 209–249. - PubMed
-
- Ajani JA, D’Amico TA, Bentrem DJ, et al.. Gastric cancer, Version 2.2022, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw 2022; 20: 167–192. - PubMed
-
- Cunningham D, Starling N, Rao S, et al.. Capecitabine and oxaliplatin for advanced esophagogastric cancer. N Engl J Med 2008; 358: 36–46. - PubMed
-
- Koizumi W, Narahara H, Hara T, et al.. S-1 plus cisplatin versus S-1 alone for first-line treatment of advanced gastric cancer (SPIRITS trial): a phase III trial. Lancet Oncol 2008; 9: 215–221. - PubMed
-
- Bang YJ, Van Cutsem E, Feyereislova A, et al.. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastrooesophageal junction cancer (ToGA): a phase 3, open-label, randomized controlled trial. Lancet 2010; 376: 687–697. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Research Materials

