This is a preprint.
Fibroblast Growth Factor (FGF) 23 and FGF Receptor 4 promote cardiac metabolic remodeling in chronic kidney disease
- PMID: 38196615
- PMCID: PMC10775858
- DOI: 10.21203/rs.3.rs-3705543/v1
Fibroblast Growth Factor (FGF) 23 and FGF Receptor 4 promote cardiac metabolic remodeling in chronic kidney disease
Update in
-
Fibroblast growth factor 23 and fibroblast growth factor receptor 4 promote cardiac metabolic remodeling in chronic kidney disease.Kidney Int. 2025 May;107(5):852-868. doi: 10.1016/j.kint.2025.01.024. Epub 2025 Feb 7. Kidney Int. 2025. PMID: 39923962 Free PMC article.
Abstract
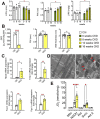
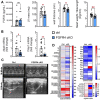
Chronic kidney disease (CKD) is a global health epidemic that significantly increases mortality due to cardiovascular disease. Left ventricular hypertrophy (LVH) is an important mechanism of cardiac injury in CKD. High serum levels of fibroblast growth factor (FGF) 23 in patients with CKD may contribute mechanistically to the pathogenesis of LVH by activating FGF receptor (FGFR) 4 signaling in cardiac myocytes. Mitochondrial dysfunction and cardiac metabolic remodeling are early features of cardiac injury that predate development of hypertrophy, but these mechanisms of disease have been insufficiently studied in models of CKD. Wild-type mice with CKD induced by adenine diet developed LVH that was preceded by morphological changes in mitochondrial structure and evidence of cardiac mitochondrial and metabolic dysfunction. In bioengineered cardio-bundles and neonatal rat ventricular myocytes grown in vitro, FGF23-mediated activation of FGFR4 caused a mitochondrial pathology, characterized by increased bioenergetic stress and increased glycolysis, that preceded the development of cellular hypertrophy. The cardiac metabolic changes and associated mitochondrial alterations in mice with CKD were prevented by global or cardiac-specific deletion of FGFR4. These findings indicate that metabolic remodeling and eventually mitochondrial dysfunction are early cardiac complications of CKD that precede structural remodeling of the heart. Mechanistically, FGF23-mediated activation of FGFR4 causes mitochondrial dysfunction, suggesting that early pharmacologic inhibition of FGFR4 might serve as novel therapeutic intervention to prevent development of LVH and heart failure in patients with CKD.
Keywords: CKD; FGF23; FGFR4; heart failure; metabolic remodeling; mitochondrial dysfunction.
Figures





References
-
- Coresh J. et al. Prevalence of chronic kidney disease in the United States. JAMA 298, 2038–2047 (2007). - PubMed
-
- Go A. S., Chertow G. M., Fan D., McCulloch C. E. & Hsu C. Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N Engl J Med 351, 1296–1305 (2004). - PubMed
-
- Weiner D. E. et al. Chronic kidney disease as a risk factor for cardiovascular disease and all-cause mortality: a pooled analysis of community-based studies. J Am Soc Nephrol 15, 1307–1315 (2004). - PubMed
-
- Schefold J. C., Filippatos G., Hasenfuss G., Anker S. D. & von Haehling S. Heart failure and kidney dysfunction: epidemiology, mechanisms and management. Nat Rev Nephrol 12, 610–623 (2016). - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

