Absent in Melanoma (AIM)2 Promotes the Outcome of Islet Transplantation by Repressing Ischemia-Induced Interferon (IFN) Signaling
- PMID: 38201220
- PMCID: PMC10778091
- DOI: 10.3390/cells13010016
Absent in Melanoma (AIM)2 Promotes the Outcome of Islet Transplantation by Repressing Ischemia-Induced Interferon (IFN) Signaling
Abstract
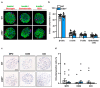
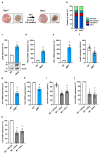
Clinical islet transplantation is limited by ischemia-induced islet cell death. Recently, it has been reported that the absent in melanoma (AIM)2 inflammasome is upregulated by ischemic cell death due to recognition of aberrant cytoplasmic self-dsDNA. However, it is unknown whether AIM2 determines the outcome of islet transplantation. To investigate this, isolated wild type (WT) and AIM2-deficient (AIM2-/-) islets were exposed to oxygen-glucose deprivation to mimic ischemia, and their viability, endocrine function, and interferon (IFN) signaling were assessed. Moreover, the revascularization and endocrine function of grafted WT and AIM2-/- islets were analyzed in the mouse dorsal skinfold chamber model and the diabetic kidney capsule model. Ischemic WT and AIM2-/- islets did not differ in their viability. However, AIM2-/- islets exhibited a higher protein level of p202, a transcriptional regulator of IFN-β and IFN-γ gene expression. Accordingly, these cytokines were upregulated in AIM2-/- islets, resulting in a suppressed gene expression and secretion of insulin. Moreover, the revascularization of AIM2-/- islet grafts was deteriorated when compared to WT controls. Furthermore, transplantation of AIM2-/- islets in diabetic mice failed to restore physiological blood glucose levels. These findings indicate that AIM2 crucially determines the engraftment and endocrine function of transplanted islets by repressing IFN signaling.
Keywords: AIM2; inflammasome; interferon; islet transplantation; revascularization.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


Similar articles
-
Targeting Pancreatic Islet NLRP3 Improves Islet Graft Revascularization.Diabetes. 2022 Aug 1;71(8):1706-1720. doi: 10.2337/db21-0851. Diabetes. 2022. PMID: 35622000
-
The role of interferon regulatory factor-1 in cytokine-induced mRNA expression and cell death in murine pancreatic beta-cells.Eur Cytokine Netw. 1999 Sep;10(3):403-12. Eur Cytokine Netw. 1999. PMID: 10477397
-
Impact of donor and prolonged cold ischemia time of neonatal pig pancreas on neonatal pig islet transplant outcome.Xenotransplantation. 2021 Mar;28(2):e12663. doi: 10.1111/xen.12663. Epub 2020 Nov 23. Xenotransplantation. 2021. PMID: 33230864
-
Absent in Melanoma 2 proteins in SLE.Clin Immunol. 2017 Mar;176:42-48. doi: 10.1016/j.clim.2016.12.011. Epub 2017 Jan 3. Clin Immunol. 2017. PMID: 28062222 Free PMC article. Review.
-
The Role of the AIM2 Gene in Obesity-Related Glucose and Lipid Metabolic Disorders: A Recent Update.Diabetes Metab Syndr Obes. 2024 Oct 21;17:3903-3916. doi: 10.2147/DMSO.S488978. eCollection 2024. Diabetes Metab Syndr Obes. 2024. PMID: 39465122 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

