CREB Is Indispensable to KIT Function in Human Skin Mast Cells-A Positive Feedback Loop between CREB and KIT Orchestrates Skin Mast Cell Fate
- PMID: 38201246
- PMCID: PMC10778115
- DOI: 10.3390/cells13010042
CREB Is Indispensable to KIT Function in Human Skin Mast Cells-A Positive Feedback Loop between CREB and KIT Orchestrates Skin Mast Cell Fate
Abstract
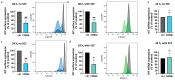
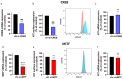
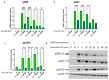
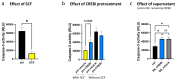
Skin mast cells (MCs) are critical effector cells in acute allergic reactions, and they contribute to chronic dermatoses like urticaria and atopic and contact dermatitis. KIT represents the cells' crucial receptor tyrosine kinase, which orchestrates proliferation, survival, and functional programs throughout the lifespan. cAMP response element binding protein (CREB), an evolutionarily well-conserved transcription factor (TF), regulates multiple cellular programs, but its function in MCs is poorly understood. We recently reported that CREB is an effector of the SCF (Stem Cell Factor)/KIT axis. Here, we ask whether CREB may also act upstream of KIT to orchestrate its functioning. Primary human MCs were isolated from skin and cultured in SCF+IL-4 (Interleukin-4). Pharmacological inhibition (666-15) and RNA interference served to manipulate CREB function. We studied KIT expression using flow cytometry and RT-qPCR, KIT-mediated signaling using immunoblotting, and cell survival using scatterplot and caspase-3 activity. The proliferation and cycle phases were quantified following BrdU incorporation. Transient CREB perturbation resulted in reduced KIT expression. Conversely, microphthalmia transcription factor (MITF) was unnecessary for KIT maintenance. KIT attenuation secondary to CREB was associated with heavily impaired KIT functional outputs, like anti-apoptosis and cell cycle progression. Likewise, KIT-elicited phosphorylation of ERK1/2 (Extracellular Signal-Regulated Kinase 1/2), AKT, and STAT5 (Signal Transducer and Activator of Transcription) was substantially diminished upon CREB inhibition. Surprisingly, the longer-term interference of CREB led to complete cell elimination, in a way surpassing KIT inhibition. Collectively, we reveal CREB as non-redundant in MCs, with its absence being incompatible with skin MCs' existence. Since SCF/KIT regulates CREB activity and, vice versa, CREB is required for KIT function, a positive feedforward loop between these elements dictates skin MCs' fate.
Keywords: CREB; KIT; SCF; apoptosis; cell cycle; mast cells; proliferation; skin; survival; transcription factors.
Conflict of interest statement
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures


Similar articles
-
CREB Is Critically Implicated in Skin Mast Cell Degranulation Elicited via FcεRI and MRGPRX2.Cells. 2024 Oct 11;13(20):1681. doi: 10.3390/cells13201681. Cells. 2024. PMID: 39451199 Free PMC article.
-
CREB Is Activated by the SCF/KIT Axis in a Partially ERK-Dependent Manner and Orchestrates Survival and the Induction of Immediate Early Genes in Human Skin Mast Cells.Int J Mol Sci. 2023 Feb 18;24(4):4135. doi: 10.3390/ijms24044135. Int J Mol Sci. 2023. PMID: 36835547 Free PMC article.
-
The SCF/KIT axis in human mast cells: Capicua acts as potent KIT repressor and ERK predominates PI3K.Allergy. 2022 Nov;77(11):3337-3349. doi: 10.1111/all.15396. Epub 2022 Jun 14. Allergy. 2022. PMID: 35652819
-
Signal transduction-associated and cell activation-linked antigens expressed in human mast cells.Int J Hematol. 2002 May;75(4):357-62. doi: 10.1007/BF02982124. Int J Hematol. 2002. PMID: 12041664 Review.
-
Regulation of mast cell phenotype by MITF.Int Arch Allergy Immunol. 2002 Feb;127(2):106-9. doi: 10.1159/000048178. Int Arch Allergy Immunol. 2002. PMID: 11919417 Review.
Cited by
-
CREB Is Critically Implicated in Skin Mast Cell Degranulation Elicited via FcεRI and MRGPRX2.Cells. 2024 Oct 11;13(20):1681. doi: 10.3390/cells13201681. Cells. 2024. PMID: 39451199 Free PMC article.
-
cAMP response element-binding protein: A credible cancer drug target.J Pharmacol Exp Ther. 2025 Apr;392(4):103529. doi: 10.1016/j.jpet.2025.103529. Epub 2025 Mar 4. J Pharmacol Exp Ther. 2025. PMID: 40157009 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

