Clinical Remission in Patients Affected by Severe Eosinophilic Asthma on Dupilumab Therapy: A Long-Term Real-Life Study
- PMID: 38202298
- PMCID: PMC10780210
- DOI: 10.3390/jcm13010291
Clinical Remission in Patients Affected by Severe Eosinophilic Asthma on Dupilumab Therapy: A Long-Term Real-Life Study
Abstract
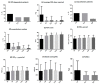
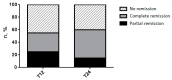
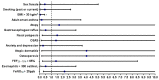
Background. Nowadays, highly selective biological drugs offer the possibility of treating severe type 2 asthma. However, in the real-life setting, it is crucial to confirm the validity of the chosen biological treatment by evaluating the achievement of clinical remission. Study purpose. The main aims of this real-life study were to evaluate the efficacy of dupilumab in terms of clinical, functional, and inflammatory outcomes at 6, 12, 18, and 24 months of treatment and to estimate the percentage of patients achieving partial or complete clinical remission at 12 and 24 months of treatment. In addition, we attempted to identify whether baseline clinical characteristics of patients could be associated with clinical remission at 24 months of treatment. Materials and methods. In this observational prospective study, 20 outpatients with severe uncontrolled eosinophilic asthma were prescribed dupilumab and followed-up after 6, 12, 18, and 24 months of treatment. At each patient visit, the need for oral corticosteroids (OCS) and corticosteroid required dose, number of exacerbations during the previous year or from the previous visit, asthma control test (ACT) score, pre-bronchodilator forced expiratory volume in the 1st second (FEV1), fractional exhaled nitric oxide at a flow rate of 50 mL/s (FeNO50), and blood eosinophil count were assessed. Results. The number of OCS-dependent patients was reduced from 10 (50%) at baseline to 5 (25%) at one year (T12) and 2 years (T24). The average dose of OCS required by patients demonstrated a significant reduction at T12 (12.5 ± 13.75 mg vs. 2.63 ± 3.94 mg, p = 0.015), remaining significant even at T24 (12.5 ± 13.75 mg vs. 2.63 ± 3.94 mg, p = 0.016). The number of exacerbators showed a statistically significant decrease at T24 (10 patients, 50% vs. 3 patients, 15%, p = 0.03). The mean number of exacerbations demonstrated a statistically significant reduction at T24 (1.45 ± 1.58 vs. 0.25 ± 0.43, p = 0.02). The ACT score improved in a statistically significant manner at T12 (15.30 ± 4.16 vs. 21.40 ± 2.35, p < 0.0001), improving further at T24 (15.30 ± 4.16 vs. 22.10 ± 2.59, p < 0.0001). The improvement in pre-bronchodilator FEV1 values reached statistical significance at T24 (79.5 ± 14.4 vs. 87.7 ± 13.8, p = 0.03). The reduction in flow at the level of the small airways (FEF25-75%) also demonstrated an improvement, although it did not reach statistical significance either at T12 or T24. A total of 11 patients (55%) showed clinical remission at T12 (6 complete + 5 partial) and 12 patients (60%) reached clinical remission at T24 (9 complete + 3 partial). Only obesity was associated with a negative odds ratio (OR) for achieving clinical remission at T24 (OR: 0.03, 95% CI: 0.002-0.41, p = 0.004). No other statistically significant differences in baseline characteristics emerged between patients who reached clinical remission at T24 and the group of patients who did not achieve this outcome. Conclusion. Dupilumab appears to be an effective drug in promoting achievement of clinical remission in patients with severe uncontrolled eosinophilic asthma. The achievement of clinical remission should be continuously evaluated during treatment. Further studies are needed to clarify whether certain baseline clinical characteristics can help predict dupilumab favorable outcomes.
Keywords: biologic therapy; clinical remission; dupilumab; severe asthma.
Conflict of interest statement
The authors declare that they have no conflicts of interest.
Figures
Similar articles
-
Younger severe asthma patients with interleukin 4 (CC variant) and dupilumab treatment are more likely to achieve clinical remission.BMC Pulm Med. 2025 Mar 21;25(1):131. doi: 10.1186/s12890-025-03578-0. BMC Pulm Med. 2025. PMID: 40119276 Free PMC article.
-
Long-term effects of dupilumab on chronic rhinosinusitis with nasal polyps: A step towards clinical remission.World Allergy Organ J. 2025 Jan 16;18(2):101024. doi: 10.1016/j.waojou.2024.101024. eCollection 2025 Feb. World Allergy Organ J. 2025. PMID: 39902112 Free PMC article.
-
Long-Term Clinical Remission on Benralizumab Treatment in Severe Eosinophilic Asthma: A Four-Year Real-Life Study.J Clin Med. 2025 Mar 18;14(6):2075. doi: 10.3390/jcm14062075. J Clin Med. 2025. PMID: 40142883 Free PMC article.
-
Matching-adjusted comparison of oral corticosteroid reduction in asthma: Systematic review of biologics.Clin Exp Allergy. 2020 Apr;50(4):442-452. doi: 10.1111/cea.13561. Epub 2020 Mar 20. Clin Exp Allergy. 2020. PMID: 31943429 Free PMC article.
-
Updated Evaluation of Dupilumab in the Treatment of Asthma: Patient Selection and Reported Outcomes.Ther Clin Risk Manag. 2020 Mar 4;16:181-187. doi: 10.2147/TCRM.S192392. eCollection 2020. Ther Clin Risk Manag. 2020. PMID: 32184610 Free PMC article. Review.
Cited by
-
Obesity-related Asthma: A Pathobiology-based Overview of Existing and Emerging Treatment Approaches.Am J Respir Crit Care Med. 2024 Nov 15;210(10):1186-1200. doi: 10.1164/rccm.202406-1166SO. Am J Respir Crit Care Med. 2024. PMID: 39311907 Review.
-
Clinical Remission Predictors in Non-Colonized Bronchiectasis and Severe Asthma with Type 2-Targeted Biologic Therapy: A Retrospective Real-Life Pilot Study.J Clin Med. 2024 Oct 22;13(21):6309. doi: 10.3390/jcm13216309. J Clin Med. 2024. PMID: 39518449 Free PMC article.
-
Patient response and remission in respiratory disease: Special focus on severe asthma and chronic obstructive pulmonary disease.J Int Med Res. 2025 May;53(5):3000605251340894. doi: 10.1177/03000605251340894. Epub 2025 May 20. J Int Med Res. 2025. PMID: 40391556 Free PMC article. Review.
-
Long-term outcomes of dupilumab therapy in severe asthma: A retrospective, multicenter, real-world study.J Allergy Clin Immunol Glob. 2025 Jul 8;4(4):100533. doi: 10.1016/j.jacig.2025.100533. eCollection 2025 Nov. J Allergy Clin Immunol Glob. 2025. PMID: 40810090 Free PMC article.
-
Obesity: Next game changer of allergic airway diseases?Clin Transl Med. 2025 May;15(5):e70316. doi: 10.1002/ctm2.70316. Clin Transl Med. 2025. PMID: 40329860 Free PMC article. Review.
References
-
- 2023 GINA Main Report—Global Initiative for Asthma—GINA. [(accessed on 18 October 2023)]. Available online: https://ginasthma.org/2023-gina-main-report/
LinkOut - more resources
Full Text Sources

