Impact of Sodium Nitroprusside on the Photosynthetic Performance of Maize and Sorghum
- PMID: 38202426
- PMCID: PMC10781006
- DOI: 10.3390/plants13010118
Impact of Sodium Nitroprusside on the Photosynthetic Performance of Maize and Sorghum
Abstract
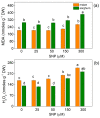
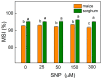
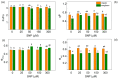
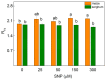
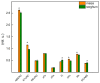
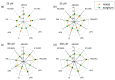
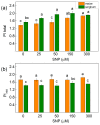
Nitric oxide (NO) is an important molecule in regulating plant growth, development and photosynthetic performance. This study investigates the impact of varying concentrations (0-300 µM) of sodium nitroprusside (SNP, a donor of NO) on the functions of the photosynthetic apparatus in sorghum (Sorghum bicolor L. Albanus) and maize (Zea mays L. Kerala) under physiological conditions. Analysis of the chlorophyll fluorescence signals (using PAM and the JIP-test) revealed an increased amount of open PSII reaction centers (qP increased), but it did not affect the number of active reaction centers per PSII antenna chlorophyll (RC/ABS). In addition, the smaller SNP concentrations (up to 150 μM) alleviated the interaction of QA with plastoquine in maize, while at 300 μM it predominates the electron recombination on QAQB-, with the oxidized S2 (or S3) states of oxygen evolving in complex ways in both studied plant species. At the same time, SNP application stimulated the electron flux-reducing end electron acceptors at the PSI acceptor side per reaction center (REo/RC increased up to 26%) and the probability of their reduction (φRo increased up to 20%). An increase in MDA (by about 30%) and H2O2 contents was registered only at the highest SNP concentration (300 µM). At this concentration, SNP differentially affected the amount of P700+ in studied plant species, i.e., it increased (by 10%) in maize but decreased (by 16%) in sorghum. The effects of SNP on the functions of the photosynthetic apparatus were accompanied by an increase in carotenoid content in both studied plants. Additionally, data revealed that SNP-induced changes in the photosynthetic apparatus differed between maize and sorghum, suggesting species specificity for SNP's impact on plants.
Keywords: JIP parameters; chlorophyll fluorescence; nitric oxide; photosynthetic performance; photosystem I; photosystem II.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Exploring Nitric Oxide as a Regulator in Salt Tolerance: Insights into Photosynthetic Efficiency in Maize.Plants (Basel). 2024 May 10;13(10):1312. doi: 10.3390/plants13101312. Plants (Basel). 2024. PMID: 38794383 Free PMC article.
-
Protective Effects of Sodium Nitroprusside on Photosynthetic Performance of Sorghum bicolor L. under Salt Stress.Plants (Basel). 2023 Feb 13;12(4):832. doi: 10.3390/plants12040832. Plants (Basel). 2023. PMID: 36840183 Free PMC article.
-
Different Sensitivity Levels of the Photosynthetic Apparatus in Zea mays L. and Sorghum bicolor L. under Salt Stress.Plants (Basel). 2021 Jul 17;10(7):1469. doi: 10.3390/plants10071469. Plants (Basel). 2021. PMID: 34371672 Free PMC article.
-
Assessment of the Photosynthetic Apparatus Functions by Chlorophyll Fluorescence and P700 Absorbance in C3 and C4 Plants under Physiological Conditions and under Salt Stress.Int J Mol Sci. 2022 Mar 29;23(7):3768. doi: 10.3390/ijms23073768. Int J Mol Sci. 2022. PMID: 35409126 Free PMC article.
-
Sensitivity of the Photosynthetic Apparatus in Maize and Sorghum under Different Drought Levels.Plants (Basel). 2023 Apr 30;12(9):1863. doi: 10.3390/plants12091863. Plants (Basel). 2023. PMID: 37176921 Free PMC article.
Cited by
-
Nitric Oxide and Photosynthesis Interplay in Plant Interactions with Pathogens.Int J Mol Sci. 2025 Jul 20;26(14):6964. doi: 10.3390/ijms26146964. Int J Mol Sci. 2025. PMID: 40725211 Free PMC article. Review.
-
Exploring Nitric Oxide as a Regulator in Salt Tolerance: Insights into Photosynthetic Efficiency in Maize.Plants (Basel). 2024 May 10;13(10):1312. doi: 10.3390/plants13101312. Plants (Basel). 2024. PMID: 38794383 Free PMC article.
-
Nitric Oxide Mitigates the Deleterious Effects Caused by Infection of Pseudomonas syringae pv. syringae and Modulates the Carbon Assimilation Process in Sweet Cherry under Water Stress.Plants (Basel). 2024 May 14;13(10):1361. doi: 10.3390/plants13101361. Plants (Basel). 2024. PMID: 38794433 Free PMC article.
References
-
- Misra A.N., Singh R., Misra M., Vladkova R., Dobrikova A.G., Apostolova E.L. Photosynthesis: Structures, Mechanisms, and Applications. Springer International Publishing; Cham, Switzerland: 2017. Nitric Oxide Mediated Effects on Chloroplasts; pp. 305–320.
-
- Ferreira L.C., Cataneo A.C. Nitric oxide in plants: A brief discussion on this multifunctional molecule. Sci. Agric. 2010;67:236–243. doi: 10.1590/S0103-90162010000200017. - DOI
-
- Gao Z., Zhang J., Zhang J., Zhang W., Zheng L., Borjigin T., Wang Y. Nitric oxide alleviates salt-induced stress damage by regulating the ascorbate–glutathione cycle and Na+/K+ homeostasis in Nitraria tangutorum Bobr. Plant Physiol. Biochem. 2022;173:46–58. doi: 10.1016/j.plaphy.2022.01.017. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

