Resonance-Based Sensing of Magnetic Nanoparticles Using Microfluidic Devices with Ferromagnetic Antidot Nanostructures
- PMID: 38202474
- PMCID: PMC10780436
- DOI: 10.3390/nano14010019
Resonance-Based Sensing of Magnetic Nanoparticles Using Microfluidic Devices with Ferromagnetic Antidot Nanostructures
Abstract
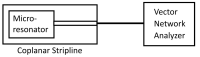
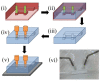
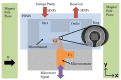
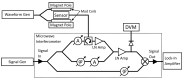
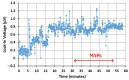
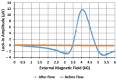
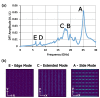
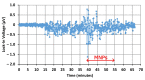
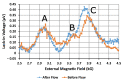
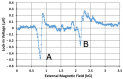
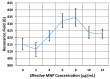
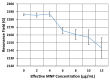
We demonstrated resonance-based detection of magnetic nanoparticles employing novel designs based upon planar (on-chip) microresonators that may serve as alternatives to conventional magnetoresistive magnetic nanoparticle detectors. We detected 130 nm sized magnetic nanoparticle clusters immobilized on sensor surfaces after flowing through PDMS microfluidic channels molded using a 3D printed mold. Two detection schemes were investigated: (i) indirect detection incorporating ferromagnetic antidot nanostructures within microresonators, and (ii) direct detection of nanoparticles without an antidot lattice. Using scheme (i), magnetic nanoparticles noticeably downshifted the resonance fields of an antidot nanostructure by up to 207 G. In a similar antidot device in which nanoparticles were introduced via droplets rather than a microfluidic channel, the largest shift was only 44 G with a sensitivity of 7.57 G/ng. This indicated that introduction of the nanoparticles via microfluidics results in stronger responses from the ferromagnetic resonances. The results for both devices demonstrated that ferromagnetic antidot nanostructures incorporated within planar microresonators can detect nanoparticles captured from dispersions. Using detection scheme (ii), without the antidot array, we observed a strong resonance within the nanoparticles. The resonance's strength suggests that direct detection is more sensitive to magnetic nanoparticles than indirect detection using a nanostructure, in addition to being much simpler.
Keywords: ferromagnetic materials; microfluidics; soft lithography; superparamagnetic iron-oxide nanoparticles.
Conflict of interest statement
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures




Similar articles
-
Controlled Capture of Magnetic Nanoparticles from Microfluidic Flows by Ferromagnetic Antidot and Dot Nanostructures.Nanomaterials (Basel). 2025 Jan 16;15(2):132. doi: 10.3390/nano15020132. Nanomaterials (Basel). 2025. PMID: 39852747 Free PMC article.
-
Application of Magnonic Crystals in Magnetic Bead Detection.Nanomaterials (Basel). 2022 Sep 21;12(19):3278. doi: 10.3390/nano12193278. Nanomaterials (Basel). 2022. PMID: 36234407 Free PMC article.
-
Magnetic force-based multiplexed immunoassay using superparamagnetic nanoparticles in microfluidic channel.Lab Chip. 2005 Jun;5(6):657-64. doi: 10.1039/b502225h. Epub 2005 Apr 29. Lab Chip. 2005. PMID: 15915258
-
Microfluidic Synthesis, Control, and Sensing of Magnetic Nanoparticles: A Review.Micromachines (Basel). 2021 Jun 29;12(7):768. doi: 10.3390/mi12070768. Micromachines (Basel). 2021. PMID: 34210058 Free PMC article. Review.
-
Magnetic nanoparticles in microfluidic and sensing: From transport to detection.Electrophoresis. 2020 Jul;41(13-14):1206-1224. doi: 10.1002/elps.201900377. Epub 2020 May 17. Electrophoresis. 2020. PMID: 32347555 Review.
Cited by
-
Observation of Linear Magnetoresistance in MoO2.Nanomaterials (Basel). 2024 May 23;14(11):915. doi: 10.3390/nano14110915. Nanomaterials (Basel). 2024. PMID: 38869538 Free PMC article.
References
-
- Hasany S.F., Rehman A., Jose R., Ahmed I. Iron oxide magnetic nanoparticles: A short review. AIP Conf. Proc. 2012;1502:298–321. doi: 10.1063/1.4769153. - DOI
-
- Wu K., Su D., Saha R., Wong D., Wang J.-P. Magnetic particle spectroscopy-based bioassays: Methods, applications, advances, and future opportunities. J. Phys. D Appl. Phys. 2019;52:173001. doi: 10.1088/1361-6463/ab03c0. - DOI
LinkOut - more resources
Full Text Sources

