Antiallergic Activity of 3- O-Dodecyl-l-ascorbic Acid
- PMID: 38202652
- PMCID: PMC10779884
- DOI: 10.3390/molecules29010069
Antiallergic Activity of 3- O-Dodecyl-l-ascorbic Acid
Abstract
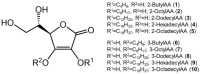
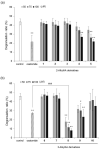
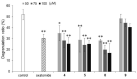
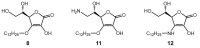
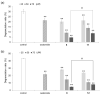
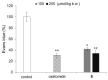
2-O-Alkyl-l-ascorbic acids and 3-O-alkyl-l-ascorbic acids were synthesized, and their degranulation inhibitory activities were evaluated. Among ascorbic acid derivatives with butyl, octyl, dodecyl, hexadecyl, and octadecyl groups introduced at the C-2 or C-3 positions, an AA derivative with a dodecyl group introduced at the C-3 position, 3-O-dodecyl-l-ascorbic acid (compound 8), showed the strongest inhibitory activity against antigen-stimulated degranulation. Compound 8 also inhibited calcium ionophore-stimulated degranulation. Compound 11, in which the hydroxyl group at the C-6 position of compound 8 was substituted with an amino group, and compound 12, in which the dodecyloxy group at the C-3 position of compound 8 was exchanged with a dodecylamino group, were synthesized, and these derivatives showed weaker inhibitory activity against antigen-stimulated degranulation than that of compound 8. In addition, orally administered compound 8 inhibited passive cutaneous anaphylaxis reactions in mice with a potency equal to that of oxatomide, an antiallergic agent. These results suggest that compound 8 may be a candidate for antiallergic treatment.
Keywords: RBL-2H3 cells; ascorbic acid derivatives; degranulation inhibitory activity; passive cutaneous anaphylaxis (PCA) reaction.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Antiallergic Activity of 6-Deoxy-2-O-methyl-6-(N-hexadecanoyl)amino-l-ascorbic Acid.Molecules. 2021 Aug 3;26(15):4684. doi: 10.3390/molecules26154684. Molecules. 2021. PMID: 34361837 Free PMC article.
-
Anti-Allergic Activity of Monoacylated Ascorbic Acid 2-Glucosides.Molecules. 2017 Dec 12;22(12):2202. doi: 10.3390/molecules22122202. Molecules. 2017. PMID: 29231858 Free PMC article.
-
Antiallergic activity of alkyl substituted pyrazolo[3, 4-d]pyrimidine (compound 88-765).Indian J Exp Biol. 1995 Jan;33(1):38-40. Indian J Exp Biol. 1995. PMID: 9135673
-
Antiallergic activity profile in vitro RBL-2H3 and in vivo passive cutaneous anaphylaxis mouse model of new sila-substituted 1,3,4-oxadiazoles.J Med Chem. 2012 Jul 26;55(14):6438-44. doi: 10.1021/jm300421h. Epub 2012 Jul 17. J Med Chem. 2012. PMID: 22770426
-
Antiallergic effect of fisetin on IgE-mediated mast cell activation in vitro and on passive cutaneous anaphylaxis (PCA).J Nutr Biochem. 2017 Oct;48:103-111. doi: 10.1016/j.jnutbio.2017.06.010. Epub 2017 Jun 30. J Nutr Biochem. 2017. PMID: 28797929
References
-
- Haahtela T., Holgate S., Pawankar R., Akdis C.A., Benjaponpitak S., Caraballo L., Demain J., Portnoy J., von Hertzen L., WAO Special Committee on Climate Change and Biodiversity The biodiversity hypothesis and allergic disease: World allergy organization position statement. World Allergy Organ. J. 2013;6:3. doi: 10.1186/1939-4551-6-3. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

