pH-Dependent HEWL-AuNPs Interactions: Optical Study
- PMID: 38202662
- PMCID: PMC10779547
- DOI: 10.3390/molecules29010082
pH-Dependent HEWL-AuNPs Interactions: Optical Study
Abstract
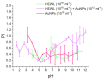
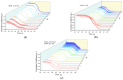
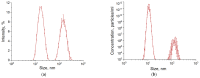
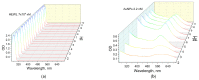
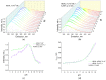
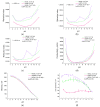
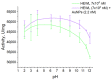
Optical methods (spectroscopy, spectrofluorometry, dynamic light scattering, and refractometry) were used to study the change in the state of hen egg-white lysozyme (HEWL), protein molecules, and gold nanoparticles (AuNPs) in aqueous colloids with changes in pH, and the interaction of protein molecules with nanoparticles was also studied. It was shown that changing pH may be the easiest way to control the protein corona on gold nanoparticles. In a colloid of nanoparticles, both in the presence and absence of protein, aggregation-deaggregation, and in a protein colloid, monomerization-dimerization-aggregation are the main processes when pH is changed. A specific point at pH 7.5, where a transition of the colloidal system from one state to another is observed, has been found using all the optical methods mentioned. It has been shown that gold nanoparticles can stabilize HEWL protein molecules at alkaline pH while maintaining enzymatic activity, which can be used in practice. The data obtained in this manuscript allow for the state of HEWL colloids and gold nanoparticles to be monitored using one or two simple and accessible optical methods.
Keywords: aggregation; hen egg-white lysozyme; nano-sized gold; nanoparticles; pH; protein.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





Similar articles
-
Proline functionalized gold nanoparticles modulates lysozyme fibrillation.Colloids Surf B Biointerfaces. 2019 Feb 1;174:401-408. doi: 10.1016/j.colsurfb.2018.11.032. Epub 2018 Nov 20. Colloids Surf B Biointerfaces. 2019. PMID: 30476794
-
Optical characterization and tunable antibacterial properties of gold nanoparticles with common proteins.Anal Biochem. 2021 Jan 1;612:113975. doi: 10.1016/j.ab.2020.113975. Epub 2020 Sep 20. Anal Biochem. 2021. PMID: 32966803
-
Sodium louroyl sarcosinate (sarkosyl) modulate amyloid fibril formation in hen egg white lysozyme (HEWL) at alkaline pH: a molecular insight study.J Biomol Struct Dyn. 2018 May;36(6):1550-1565. doi: 10.1080/07391102.2017.1329097. Epub 2017 May 28. J Biomol Struct Dyn. 2018. PMID: 28490248
-
Pharmacological Modulation of Host Immunity with Hen Egg White Lysozyme (HEWL)-A Review.Molecules. 2023 Jun 27;28(13):5027. doi: 10.3390/molecules28135027. Molecules. 2023. PMID: 37446691 Free PMC article. Review.
-
Lysozyme: a model enzyme in protein crystallography.EXS. 1996;75:185-222. doi: 10.1007/978-3-0348-9225-4_11. EXS. 1996. PMID: 8765301 Review.
References
-
- Kononenko V.V., Ashikkalieva K.K., Arutyunyan N.R., Romshin A.M., Kononenko T.V., Konov V.I. Femtosecond laser-produced plasma driven nanoparticle formation in gold aqueous solution. J. Photochem. Photobiol. A Chem. 2022;426:113709. doi: 10.1016/j.jphotochem.2021.113709. - DOI
-
- Nastulyavichus A.A., Kudryashov S.I., Emelyanenko A.M., Boinovich L.B. Laser Generation of Colloidal Nanoparticles in Liquids: Key Processes of Laser Dispersion and Main Characteristics of Nanoparticles. Colloid J. 2023;85:233–250. doi: 10.1134/S1061933X23600136. - DOI
-
- Gawas G., Ayyanar M., Gurav N., Hase D., Murade V., Nadaf S., Khan M.S., Chikhale R., Kalaskar M., Gurav S. Process Optimization for the Bioinspired Synthesis of Gold Nanoparticles Using Cordyceps militaris, Its Characterization, and Assessment of Enhanced Therapeutic Efficacy. Pharmaceuticals. 2023;16:1311. doi: 10.3390/ph16091311. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

