Diversity-Orientated Synthesis and Biological Properties of Compounds Based on the N-Phenylquinoneimine Scaffold
- PMID: 38202832
- PMCID: PMC10780808
- DOI: 10.3390/molecules29010249
Diversity-Orientated Synthesis and Biological Properties of Compounds Based on the N-Phenylquinoneimine Scaffold
Abstract
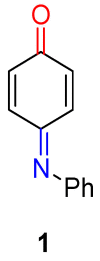
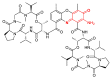
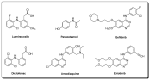
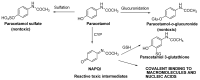
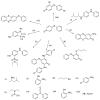
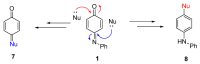
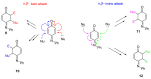
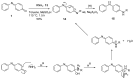
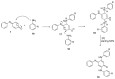
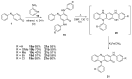
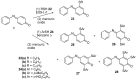
The N-phenylquinoneimine scaffold is a versatile synthetic platform that has gained significant attention in the field of drug discovery due to its structural diversity and capacity to interact with biologically relevant targets. This review explores established synthetic methodologies and highlights the significant biological activities exhibited by compounds derived from this scaffold, their implications for medicinal chemistry, and the development of novel therapeutics.
Keywords: DNA; N-phenylquinoneimine; biological activity; diversity-orientated synthesis; drugs; reactive metabolites.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
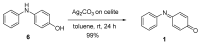
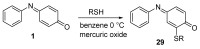

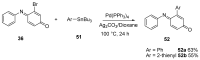
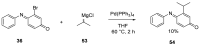
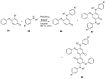
Similar articles
-
Investigation of innovative synthesis of biologically active compounds on the basis of newly developed reactions.Chem Pharm Bull (Tokyo). 2012;60(6):687-705. doi: 10.1248/cpb.60.687. Chem Pharm Bull (Tokyo). 2012. PMID: 22689419 Review.
-
Chalcone: A Privileged Structure in Medicinal Chemistry.Chem Rev. 2017 Jun 28;117(12):7762-7810. doi: 10.1021/acs.chemrev.7b00020. Epub 2017 May 10. Chem Rev. 2017. PMID: 28488435 Free PMC article. Review.
-
Pyrrolo[2,1-a]isoquinoline scaffold in drug discovery: advances in synthesis and medicinal chemistry.Future Med Chem. 2019 Oct;11(20):2735-2755. doi: 10.4155/fmc-2019-0136. Epub 2019 Sep 26. Future Med Chem. 2019. PMID: 31556691 Review.
-
Structural diversity and potency range distribution of scaffolds from compounds active against current pharmaceutical targets.Future Med Chem. 2015;7(2):111-22. doi: 10.4155/fmc.14.156. Future Med Chem. 2015. PMID: 25686001
-
An overview of spirooxindole as a promising scaffold for novel drug discovery.Expert Opin Drug Discov. 2020 May;15(5):603-625. doi: 10.1080/17460441.2020.1733526. Epub 2020 Feb 28. Expert Opin Drug Discov. 2020. PMID: 32106717 Review.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

