Recent Advances in Organometallic NIR Iridium(III) Complexes for Detection and Therapy
- PMID: 38202839
- PMCID: PMC10780525
- DOI: 10.3390/molecules29010256
Recent Advances in Organometallic NIR Iridium(III) Complexes for Detection and Therapy
Abstract
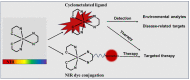
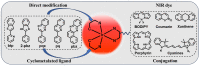
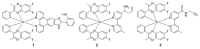
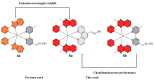
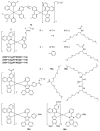
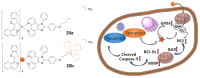
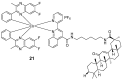
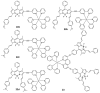
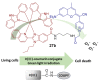
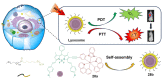
Iridium(III) complexes are emerging as a promising tool in the area of detection and therapy due to their prominent photophysical properties, including higher photostability, tunable phosphorescence emission, long-lasting phosphorescence, and high quantum yields. In recent years, much effort has been devoted to develop novel near-infrared (NIR) iridium(III) complexes to improve signal-to-noise ratio and enhance tissue penetration. In this review, we summarize different classes of organometallic NIR iridium(III) complexes for detection and therapy, including cyclometalated ligand-enabled NIR iridium(III) complexes and NIR-dye-conjugated iridium(III) complexes. Moreover, the prospects and challenges for organometallic NIR iridium(III) complexes for targeted detection and therapy are discussed.
Keywords: NIR; detection; iridium(III) complexes; therapy.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures




Similar articles
-
Group 9 organometallic compounds for therapeutic and bioanalytical applications.Acc Chem Res. 2014 Dec 16;47(12):3614-31. doi: 10.1021/ar500310z. Epub 2014 Nov 4. Acc Chem Res. 2014. PMID: 25369127
-
Near-infrared emitting iridium complexes: Molecular design, photophysical properties, and related applications.iScience. 2021 Jul 17;24(8):102858. doi: 10.1016/j.isci.2021.102858. eCollection 2021 Aug 20. iScience. 2021. PMID: 34381981 Free PMC article. Review.
-
Recent Advancement in the Synthesis of Ir-Based Complexes.ACS Omega. 2023 Nov 6;8(46):43408-43432. doi: 10.1021/acsomega.3c04867. eCollection 2023 Nov 21. ACS Omega. 2023. PMID: 38027378 Free PMC article. Review.
-
Iridium(iii) complexes with the dithieno[3,2-b:2',3'-d]phosphole oxide group and their high optical power limiting performances.Dalton Trans. 2020 Apr 15;49(15):4967-4976. doi: 10.1039/d0dt00102c. Dalton Trans. 2020. PMID: 32236211
-
Near-infrared emitting iridium(iii) complexes for mitochondrial imaging in living cells.Dalton Trans. 2017 Apr 5;46(14):4777-4785. doi: 10.1039/c7dt00255f. Dalton Trans. 2017. PMID: 28345088
Cited by
-
Development of a NIR Iridium(III) Complex-Based Probe for the Selective Detection of Iron(II) Ions.Biosensors (Basel). 2024 Jul 29;14(8):369. doi: 10.3390/bios14080369. Biosensors (Basel). 2024. PMID: 39194598 Free PMC article.
-
(2,9-Dimethyl-1,10-phenanthroline)bis-[2-(pyridin-2-yl)phen-yl]iridium(III) hexa-fluoro-phosphate and (2,9-dimethyl-1,10-phenanthroline)bis-[5-methyl-2-(pyridin-2-yl)phen-yl]iridium(III) hexa-fluoro-phosphate-diethyl ether-aceto-nitrile (1/0.61/0.78).Acta Crystallogr E Crystallogr Commun. 2025 Jan 14;81(Pt 2):127-131. doi: 10.1107/S2056989024012039. eCollection 2025 Feb 1. Acta Crystallogr E Crystallogr Commun. 2025. PMID: 39927383 Free PMC article.
References
-
- Deng Z.Q., Li H.C., Chen S., Wang N., Liu G.Y., Liu D.J., Ou W.H., Xu F.J., Wang X., Lei D.Y., et al. Near-infrared-activated anticancer platinum(IV) complexes directly photooxidize biomolecules in an oxygen-independent manner. Nat. Chem. 2023;15:930–939. doi: 10.1038/s41557-023-01242-w. - DOI - PubMed
-
- Kim H.U., Kim T., Kim C., Kim M., Park T. Recent Advances in Structural Design of Efficient Near-Infrared Light-Emitting Organic Small Molecules. Adv. Funct. Mater. 2023;33:2208082. doi: 10.1002/adfm.202208082. - DOI
-
- Martinic I., Eliseeva S.V., Petoud S. Near-infrared emitting probes for biological imaging: Organic fluorophores, quantum dots, fluorescent proteins, lanthanide(III) complexes and nanomaterials. J. Lumin. 2017;189:19–43. doi: 10.1016/j.jlumin.2016.09.058. - DOI
Publication types
MeSH terms
Substances
Grants and funding
- 22101230/National Natural Science Foundation of China
- D5000230060/Fundamental Research Funds for the Central Universities
- 21YF1451200/Shanghai Sailing Program
- cstc2021jcyj-msxm2073/Natural Science Foundation of Chongqing, China
- 22JHQ082/Shaanxi Fundamental Science Re-search Project for Chemistry & Biology
- 2021A1515110840, 2023A1515011871/Guangdong Basic and Applied Basic Research Foundation
- 0007/2020/A1, 0020/2022/A1/Science and Technology Development Fund
- SKL-QRCM(UM)-2023-2025, SKL-QRCM-IRG2023-025/State Key Laboratory of Quality Research in Chinese Medicine (University of Macau)
- MYRG2023-00194-ICMS, MYRG2020-00017-ICMS, MYRG2022-00137-ICMS/University of Macau
LinkOut - more resources
Full Text Sources
Miscellaneous

