Optical Coherence Tomography as a Non-Invasive Tool for Plant Material Characterization in Agriculture: A Review
- PMID: 38203080
- PMCID: PMC10781338
- DOI: 10.3390/s24010219
Optical Coherence Tomography as a Non-Invasive Tool for Plant Material Characterization in Agriculture: A Review
Abstract
Characterizing plant material is crucial in terms of early disease detection, pest control, physiological assessments, and growth monitoring, which are essential parameters to increase production in agriculture and prevent unnecessary economic losses. The conventional methods employed to assess the aforementioned parameters have several limitations, such as invasive inspection, complexity, high time consumption, and costly features. In recent years, optical coherence tomography (OCT), which is an ultra-high resolution, non-invasive, and real-time unique image-based approach has been widely utilized as a significant and potential tool for assessing plant materials in numerous aspects. The obtained OCT cross-sections and volumetrics, as well as the amplitude signals of plant materials, have the capability to reveal vital information in both axial and lateral directions owing to the high resolution of the imaging system. This review discusses recent technological trends and advanced applications of OCT, which have been potentially adapted for numerous agricultural applications, such as non-invasive disease screening, optical signals-based growth speed detection, the structural analysis of plant materials, and microbiological discoveries. Therefore, this review offers a comprehensive exploration of recent advanced OCT technological approaches for agricultural applications, which provides insights into their potential to incorporate OCT technology into numerous industries.
Keywords: agriculture; disease detection; image analysis; image processing; optical coherence tomography; optical imaging.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

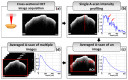
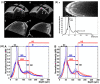
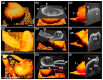

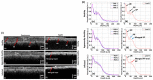



References
-
- Teng P., Krupa S.V. Assessment of losses which constrain production and crop improvement in agriculture and forestry; Proceedings of the EC Stakman Commemorative Symposium (1980: University of Minnesota); Minneapolis, MN, USA. 20–23 August 1980; Minneapolis, MN, USA: Department of Plant Pathology, University of Minnesota; 1980.
-
- Teng P.S. Crop Loss Assessment and Pest Management. American Phytopathological Society; St. Paul, MN, USA: 1987.
-
- Oerke E.-C., Dehne H.-W., Schönbeck F., Weber A. Crop Production and Crop Protection: Estimated Losses in Major Food and Cash Crops. Elsevier; Amsterdam, The Netherlands: 2012.
-
- Oerke E.-C. Crop losses to pests. J. Agric. Sci. 2006;144:31–43. doi: 10.1017/S0021859605005708. - DOI
-
- Pimentel D., Zuniga R., Morrison D. Update on the environmental and economic costs associated with alien-invasive species in the United States. Ecol. Econ. 2005;52:273–288. doi: 10.1016/j.ecolecon.2004.10.002. - DOI
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

