A Novel Role for DOC2B in Ameliorating Palmitate-Induced Glucose Uptake Dysfunction in Skeletal Muscle Cells via a Mechanism Involving β-AR Agonism and Cofilin
- PMID: 38203312
- PMCID: PMC10779393
- DOI: 10.3390/ijms25010137
A Novel Role for DOC2B in Ameliorating Palmitate-Induced Glucose Uptake Dysfunction in Skeletal Muscle Cells via a Mechanism Involving β-AR Agonism and Cofilin
Abstract
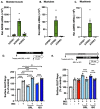
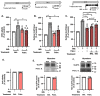
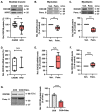
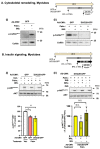
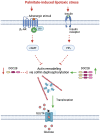
Diet-related lipotoxic stress is a significant driver of skeletal muscle insulin resistance (IR) and type 2 diabetes (T2D) onset. β2-adrenergic receptor (β-AR) agonism promotes insulin sensitivity in vivo under lipotoxic stress conditions. Here, we established an in vitro paradigm of lipotoxic stress using palmitate (Palm) in rat skeletal muscle cells to determine if β-AR agonism could cooperate with double C-2-like domain beta (DOC2B) enrichment to promote skeletal muscle insulin sensitivity under Palm-stress conditions. Previously, human T2D skeletal muscles were shown to be deficient for DOC2B, and DOC2B enrichment resisted IR in vivo. Our Palm-stress paradigm induced IR and β-AR resistance, reduced DOC2B protein levels, triggered cytoskeletal cofilin phosphorylation, and reduced GLUT4 translocation to the plasma membrane (PM). By enhancing DOC2B levels in rat skeletal muscle, we showed that the deleterious effects of palmitate exposure upon cofilin, insulin, and β-AR-stimulated GLUT4 trafficking to the PM and glucose uptake were preventable. In conclusion, we revealed a useful in vitro paradigm of Palm-induced stress to test for factors that can prevent/reverse skeletal muscle dysfunctions related to obesity/pre-T2D. Discerning strategies to enrich DOC2B and promote β-AR agonism can resist skeletal muscle IR and halt progression to T2D.
Keywords: DOC2B; GLUT4; cofilin; glucose uptake; insulin resistance; lipotoxic stress; palmitate; skeletal muscle; type 2 diabetes; β-adrenergic receptor.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
DOC2B promotes insulin sensitivity in mice via a novel KLC1-dependent mechanism in skeletal muscle.Diabetologia. 2019 May;62(5):845-859. doi: 10.1007/s00125-019-4824-2. Epub 2019 Feb 1. Diabetologia. 2019. PMID: 30707251 Free PMC article.
-
Doc2b enrichment enhances glucose homeostasis in mice via potentiation of insulin secretion and peripheral insulin sensitivity.Diabetologia. 2014 Jul;57(7):1476-84. doi: 10.1007/s00125-014-3227-7. Epub 2014 Apr 6. Diabetologia. 2014. PMID: 24705606 Free PMC article.
-
Doc2b is a key effector of insulin secretion and skeletal muscle insulin sensitivity.Diabetes. 2012 Oct;61(10):2424-32. doi: 10.2337/db11-1525. Epub 2012 Jun 14. Diabetes. 2012. PMID: 22698913 Free PMC article.
-
Protein kinase C iota facilitates insulin-induced glucose transport by phosphorylation of soluble nSF attachment protein receptor regulator (SNARE) double C2 domain protein b.J Diabetes Investig. 2019 May;10(3):591-601. doi: 10.1111/jdi.12965. Epub 2018 Dec 1. J Diabetes Investig. 2019. PMID: 30369065 Free PMC article.
-
DOC2B: a novel syntaxin-4 binding protein mediating insulin-regulated GLUT4 vesicle fusion in adipocytes.Diabetes. 2009 Feb;58(2):377-84. doi: 10.2337/db08-0303. Epub 2008 Nov 25. Diabetes. 2009. PMID: 19033398 Free PMC article.
Cited by
-
Methylglyoxal-Derived Nucleoside Adducts Drive Vascular Dysfunction in a RAGE-Dependent Manner.Antioxidants (Basel). 2024 Jan 10;13(1):85. doi: 10.3390/antiox13010085. Antioxidants (Basel). 2024. PMID: 38247509 Free PMC article.
-
Pancreatic β-cells package double C2-like domain beta protein into extracellular vesicles via tandem C2 domains.Front Endocrinol (Lausanne). 2024 Oct 21;15:1451279. doi: 10.3389/fendo.2024.1451279. eCollection 2024. Front Endocrinol (Lausanne). 2024. PMID: 39497805 Free PMC article.
References
-
- Mukaida S., Sato M., Oberg A.I., Dehvari N., Olsen J.M., Kocan M., Halls M.L., Merlin J., Sandstrom A.L., Csikasz R.I., et al. BRL37344 stimulates GLUT4 translocation and glucose uptake in skeletal muscle via β2-adrenoceptors without causing classical receptor desensitization. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2019;316:R666–R677. doi: 10.1152/ajpregu.00285.2018. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

