1,3-Dichloroadamantyl-Containing Ureas as Potential Triple Inhibitors of Soluble Epoxide Hydrolase, p38 MAPK and c-Raf
- PMID: 38203510
- PMCID: PMC10779153
- DOI: 10.3390/ijms25010338
1,3-Dichloroadamantyl-Containing Ureas as Potential Triple Inhibitors of Soluble Epoxide Hydrolase, p38 MAPK and c-Raf
Abstract
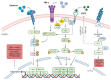
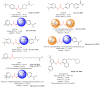
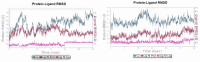
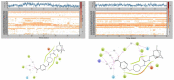
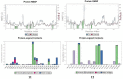
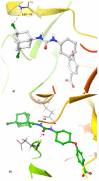
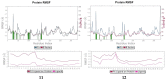
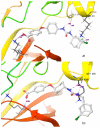
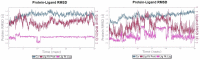
Soluble epoxide hydrolase (sEH) is an enzyme involved in the metabolism of bioactive lipid signaling molecules. sEH converts epoxyeicosatrienoic acids (EET) to virtually inactive dihydroxyeicosatrienoic acids (DHET). The first acids are "medicinal" molecules, the second increase the inflammatory infiltration of cells. Mitogen-activated protein kinases (p38 MAPKs) are key protein kinases involved in the production of inflammatory mediators, including tumor necrosis factor-α (TNF-α) and cyclooxygenase-2 (COX-2). p38 MAPK signaling plays an important role in the regulation of cellular processes, especially inflammation. The proto-oncogenic serine/threonine protein kinase Raf (c-Raf) is a major component of the mitogen-activated protein kinase (MAPK) pathway: ERK1/2 signaling. Normal cellular Raf genes can also mutate and become oncogenes, overloading the activity of MEK1/2 and ERK1/2. The development of multitarget inhibitors is a promising strategy for the treatment of socially dangerous diseases. We synthesized 1,3-disubstituted ureas and diureas containing a dichloroadamantyl moiety. The results of computational methods show that soluble epoxide hydrolase inhibitors can act on two more targets in different signaling pathways of mitogen-activated protein kinases p38 MAPK and c-Raf. The two chlorine atoms in the adamantyl moiety may provide additional Cl-π interactions in the active site of human sEH. Molecular dynamics studies have shown that the stability of ligand-protein complexes largely depends on the "spacer effect." The compound containing a bridge between the chloroadamantyl fragment and the ureide group forms more stable ligand-protein complexes with sEH and p38 MAPK, which indicates a better conformational ability of the molecule in the active sites of these targets. In turn, a compound containing two chlorine atoms forms a more stable complex with c-Raf, probably due to the presence of additional halogen bonds of chlorine atoms with amino acid residues.
Keywords: c-Raf; cancer; inhibitors; p38 MAPK; signaling pathway; soluble epoxide hydrolase (sEH); ureas.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
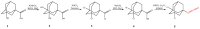




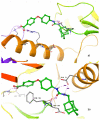







References
-
- Liu Y., Zhang Y., Schmelzer K., Lee T.S., Fang X., Zhu Y., Spector A.A., Gill S., Morisseau C., Hammock B.D., et al. The antiinflammatory effect of laminar flow: The role of PPARgamma, epoxyeicosatrienoic acids, and soluble epoxide hydrolase. Proc. Natl. Acad. Sci. USA. 2005;102:16747–16752. doi: 10.1073/pnas.0508081102. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

