Elucidating Differences in Early-Stage Centrosome Amplification in Primary and Immortalized Mouse Cells
- PMID: 38203554
- PMCID: PMC10778991
- DOI: 10.3390/ijms25010383
Elucidating Differences in Early-Stage Centrosome Amplification in Primary and Immortalized Mouse Cells
Abstract
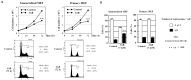
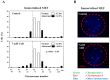
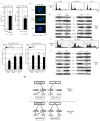
The centrosome is involved in cytoplasmic microtubule organization during interphase and in mitotic spindle assembly during cell division. Centrosome amplification (abnormal proliferation of centrosome number) has been observed in several types of cancer and in precancerous conditions. Therefore, it is important to elucidate the mechanism of centrosome amplification in order to understand the early stage of carcinogenesis. Primary cells could be used to better understand the early stage of carcinogenesis rather than immortalized cells, which tend to have various genetic and epigenetic changes. Previously, we demonstrated that a poly(ADP-ribose) polymerase (PARP) inhibitor, 3-aminobenzamide (3AB), which is known to be nontoxic and nonmutagenic, could induce centrosome amplification and chromosomal aneuploidy in CHO-K1 cells. In this study, we compared primary mouse embryonic fibroblasts (MEF) and immortalized MEF using 3AB. Although centrosome amplification was induced with 3AB treatment in immortalized MEF, a more potent PARP inhibitor, AG14361, was required for primary MEF. However, after centrosome amplification, neither 3AB in immortalized MEF nor AG14361 in primary MEF caused chromosomal aneuploidy, suggesting that further genetic and/or epigenetic change(s) are required to exhibit aneuploidy. The DNA-damaging agents doxorubicin and γ-irradiation can cause cancer and centrosome amplification in experimental animals. Although doxorubicin and γ-irradiation induced centrosome amplification and led to decreased p27Kip protein levels in immortalized MEF and primary MEF, the phosphorylation ratio of nucleophosmin (Thr199) increased in immortalized MEF, whereas it decreased in primary MEF. These results suggest that there exists a yet unidentified pathway, different from the nucleophosmin phosphorylation pathway, which can cause centrosome amplification in primary MEF.
Keywords: DNA damage (doxorubicin, irradiation); PARP inhibition; cell differentiation; centrosome amplification; chromosome instability.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

Similar articles
-
Novel pathway of centrosome amplification that does not require DNA lesions.Cancer Sci. 2012 Feb;103(2):191-6. doi: 10.1111/j.1349-7006.2011.02152.x. Epub 2011 Dec 15. Cancer Sci. 2012. PMID: 22085410
-
PARP Inhibitor Decreases Akt Phosphorylation and Induces Centrosome Amplification and Chromosomal Aneuploidy in CHO-K1 Cells.Int J Mol Sci. 2022 Mar 23;23(7):3484. doi: 10.3390/ijms23073484. Int J Mol Sci. 2022. PMID: 35408845 Free PMC article.
-
The novel poly(ADP-Ribose) polymerase inhibitor, AG14361, sensitizes cells to topoisomerase I poisons by increasing the persistence of DNA strand breaks.Clin Cancer Res. 2005 Dec 1;11(23):8449-57. doi: 10.1158/1078-0432.CCR-05-1224. Clin Cancer Res. 2005. PMID: 16322308
-
Deregulation of the centrosome cycle and the origin of chromosomal instability in cancer.Adv Exp Med Biol. 2005;570:393-421. doi: 10.1007/1-4020-3764-3_14. Adv Exp Med Biol. 2005. PMID: 18727509 Review.
-
Centrosome amplification, chromosomal instability and cancer: mechanistic, clinical and therapeutic issues.Chromosome Res. 2016 Jan;24(1):105-26. doi: 10.1007/s10577-015-9505-5. Chromosome Res. 2016. PMID: 26645976 Review.
Cited by
-
CDK2 inhibition produces a persistent population of polyploid cancer cells.JCI Insight. 2025 Apr 15;10(10):e189901. doi: 10.1172/jci.insight.189901. eCollection 2025 May 22. JCI Insight. 2025. PMID: 40232858 Free PMC article.
-
Investigation of c-Fos/c-Jun Signaling Pathways in Periostracum Cicadae's Inhibition of EMT in Gastric Tissue.Pharmaceuticals (Basel). 2025 Apr 7;18(4):537. doi: 10.3390/ph18040537. Pharmaceuticals (Basel). 2025. PMID: 40283972 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

