Comparative In Vitro Dissolution Assessment of Calcined and Uncalcined Hydroxyapatite Using Differences in Bioresorbability and Biomineralization
- PMID: 38203791
- PMCID: PMC10779548
- DOI: 10.3390/ijms25010621
Comparative In Vitro Dissolution Assessment of Calcined and Uncalcined Hydroxyapatite Using Differences in Bioresorbability and Biomineralization
Abstract
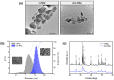
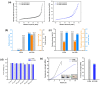
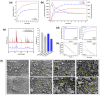
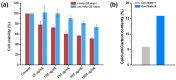
This study reports the effect of the not-calcining process on the bioresorption and biomineralization of hydroxyapatite through in vitro dissolution assessment. The prepared calcined hydroxyapatite (c-HAp) and uncalcined hydroxyapatite (unc-HAp) have a particle size of 2 μm and 13 μm, surface areas of 4.47 m2/g and 108.08 m2/g, and a Ca/P ratio of 1.66 and 1.52, respectively. In vitro dissolution assessments of c-HAp and unc-HAp were performed for 20 days at 37 °C in a citric acid buffer according to ISO 10993-14. During the dissolution, the c-HAp and unc-HAp confirmed an increase in weight, and the calcium and phosphorous ions were rapidly released. The calcium ions released from c-HAp formed rod-shaped particles with a longer and thinner morphology, while in unc-HAp, they appeared thicker and shorter. In the ICP-OES results, the concentrations of calcium elements were initially increased and then decreased by this formation. The rod-shaped particles identified as calcium citrate (Ca-citrate) through the XRD pattern. The calcium content of Ca-citrate particles from unc-HAp was higher than that from c-HAp. The unc-HAp demonstrated non-toxic properties in a cytotoxicity evaluation. Therefore, due to its higher bioresorption and biomineralization, unc-HAp exhibits enhanced biocompatibility compared to c-HAp.
Keywords: biomineralization; bioresorption; calcium citrate; dissolution; uncalcined hydroxyapatite.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


References
-
- Pramanik S., Agarwal A.K., Rai K.N., Garg A. Development of high strength hydroxyapatite by solid-state-sintering process. Ceram. Int. 2007;33:419–426. doi: 10.1016/j.ceramint.2005.10.025. - DOI
-
- Koonawoot R., Saelee C., Thiansem S., Punyanitya S. Synthesis control and characterization of hydroxyapatite ceramic using a solid state reaction; Proceedings of the 1st Mae Fah Luang University International Conference; Chiang Rai, Thailand. 29 November–1 December 2012.
-
- Cai S., Wang Y., Lv H., Peng Z., Yao K. Synthesis of carbonated hydroxyapatite nanofibers by mechanochemical methods. Ceram. Int. 2005;31:135–138. doi: 10.1016/j.ceramint.2004.04.012. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

