The AM-4 Family of Layered Titanosilicates: Single-Crystal-to-Single-Crystal Transformation, Synthesis and Ionic Conductivity
- PMID: 38203965
- PMCID: PMC10780191
- DOI: 10.3390/ma17010111
The AM-4 Family of Layered Titanosilicates: Single-Crystal-to-Single-Crystal Transformation, Synthesis and Ionic Conductivity
Abstract
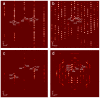
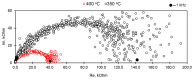
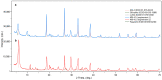
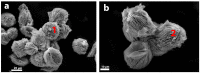
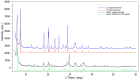
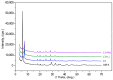
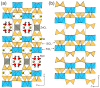
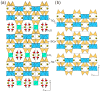
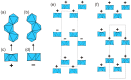
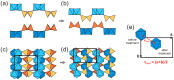
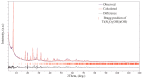
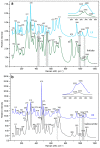
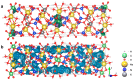
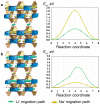
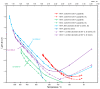
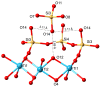
Flexible crystal() structures, which exhibit() single-crystal()-to-single-crystal() (SCSC) transformations(), are attracting attention() in many applied aspects: magnetic() switches, catalysis, ferroelectrics and sorption. Acid treatment() for titanosilicate material() AM-4 and natural() compounds with the same structures led to SCSC transformation() by loss() Na+, Li+ and Zn2+ cations with large structural() changes (20% of the unit()-cell() volume()). The conservation() of crystallinity through complex() transformation() is possible due() to the formation() of a strong hydrogen bonding() system(). The mechanism() of transformation() has been characterized using single-crystal() X-ray() diffraction analysis(), powder() diffraction, Rietvield refinement, Raman spectroscopy and electron microscopy. The low migration() energy() of cations in the considered materials() is confirmed using bond()-valence and density() functional() theory() calculations, and the ion conductivity of the AM-4 family's materials() has been experimentally verified.
Keywords: AM-4; K3; L3; SCSC; arctic minerals; impedance spectroscopy; ion-migration modeling; ionic conductivity; kukisvumite; lintisite; microporous; mineral-mimetic material; transformation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





Similar articles
-
Thermally triggered solid-state single-crystal-to-single-crystal structural transformation accompanies property changes.Chemistry. 2015 Mar 16;21(12):4703-11. doi: 10.1002/chem.201405984. Epub 2015 Feb 6. Chemistry. 2015. PMID: 25663637
-
Solvent-triggered single-crystal-to-single-crystal transformation from a monomeric to polymeric copper(II) complex based on an aza macrocyclic ligand.Acta Crystallogr B Struct Sci Cryst Eng Mater. 2020 Apr 1;76(Pt 2):225-232. doi: 10.1107/S2052520620002371. Epub 2020 Mar 19. Acta Crystallogr B Struct Sci Cryst Eng Mater. 2020. PMID: 32831224
-
Giant single-crystal-to-single-crystal transformations associated with chiral interconversion induced by elimination of chelating ligands.Nat Commun. 2021 Nov 25;12(1):6908. doi: 10.1038/s41467-021-27282-9. Nat Commun. 2021. PMID: 34824289 Free PMC article.
-
Temperature-Induced Single-Crystal-to-Single-Crystal Transformations with Consequential Changes in the Magnetic Properties of Fe(III) Complexes.ACS Omega. 2019 May 22;4(5):8731-8738. doi: 10.1021/acsomega.8b03400. eCollection 2019 May 31. ACS Omega. 2019. PMID: 31459962 Free PMC article.
-
Structural Features, Chemical Diversity, and Physical Properties of Microporous Sodalite-Type Materials: A Review.Int J Mol Sci. 2024 Sep 23;25(18):10218. doi: 10.3390/ijms251810218. Int J Mol Sci. 2024. PMID: 39337703 Free PMC article. Review.
Cited by
-
Mineral Mimetic Material Sr-Exchanged Sitinakite of Different Crystallinity: Phase Transformations during Heat Treatment and the Strength of SR Fixation in a Ceramic Matrix.Materials (Basel). 2024 Apr 25;17(9):1991. doi: 10.3390/ma17091991. Materials (Basel). 2024. PMID: 38730797 Free PMC article.
References
-
- Krivovichev S.V., editor. Minerals as Advanced Materials II. Springer; Berlin/Heidelberg, Germany: 2012.
-
- Krivovichev S.V., editor. Minerals as Advanced Materials I. Springer; Berlin/Heidelberg, Germany: 2008.
-
- Nikolaev A.I., Ivanyuk G.Y., Krivovichev S.V., Yakovenchuk V.N., Pakhomovsky Y.A., Gerasimova L.G., Maslova M.V., Selivanova E.A., Spiridonova D.V., Konopleva N.G. Nanoporous Titanosilicates: Crystallochemistry, Conditions of Localization in Alkaline Arrays, and Prospects for Synthesis. Vestn. KSC. 2010:51–62. (In Russian)
-
- Clearfield A., Bortun L., Bortun A. Alkali Metal Ion Exchange by the Framework Titanium Silicate M2Ti2O3SiO4·nH2O (M=H, Na) React. Funct. Polym. 2000;43:85–95. doi: 10.1016/S1381-5148(99)00005-X. - DOI
-
- Al-Attar L., Dyer A., Harjulab R. Sorption of Uranium onto Titanosilicate Materials. J. Radioanal. Nucl. Chem. 2001;247:121–128. doi: 10.1023/A:1006758929946. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources

