Effects of adipose-derived mesenchymal stem cell conditioned medium on human tenocytes exposed to high glucose
- PMID: 38204801
- PMCID: PMC10775729
- DOI: 10.1177/1759720X231214903
Effects of adipose-derived mesenchymal stem cell conditioned medium on human tenocytes exposed to high glucose
Abstract
Introduction: Diabetic tendinopathy is a common invalidating and challenging disease that may be treated using stem cells. However, the effects of adipose-derived mesenchymal stem cell conditioned medium (ASC-CM) in diabetic tendinopathy have never been explored.
Objectives: The present study evaluated the effects of ASC-CM on morphology, cell viability, structure, and scratch wound closure of human tenocytes (HTNC) exposed to high glucose (HG).
Design: Experimental study.
Methods: HTNC were exposed to HG (25 mM) for 7, 14 and 21 days with or without ASC-CM for the last 24 h. CM was collected from 4 × 105 ASCs, centrifuged for 10 min at 200 g and sterilized with 0.22 μm syringe filter.
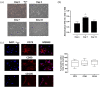
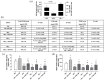
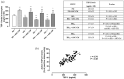
Results: At 7 days, HG-HTNC had decreased cell viability [72 ± 2%, p < 0.01 versus normal glucose (NG)] compared to NG-HTNC (90 ± 5%). A further decrement was detected after 14 and 21 days (60 ± 4% and 60 ± 5%, both, p < 0.01 versus NG and p < 0.01 versus HG7). While NG-HTNC evidenced a normal fibroblast cell-like elongated morphology, HG-HTNC showed increased cell roundness. In contrast, HG-HTNC exposed to ASC-CM showed a significant increase in cell viability, an improved cell morphology and higher scratch wound closure at all HG time points. Moreover, the exposure to ASC-CM significantly increased thrombospondin 1 and transforming growth factor beta 1 (TGF-β1) content in HG-HTNC. The TGF-β1 elevation was paralleled by higher Collagen I and Vascular Endothelial Growth Factor in HG-HTNC.
Conclusion: ASC-CM may restore the natural morphology, cell viability and structure of HTNC, promoting their scratch wound closure through TGF-β1 increase.
Keywords: adipose-derived mesenchymal stem cells; conditioned medium; diabetes; tendinopathy; thrombospondin-1; transforming growth factor beta 1.
© The Author(s), 2024.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Figures


Similar articles
-
New Insights on the miRNA Role in Diabetic Tendinopathy: Adipose-Derived Mesenchymal Stem Cell Conditioned Medium as a Potential Innovative Epigenetic-Based Therapy for Tendon Healing.Biomolecules. 2025 Feb 11;15(2):264. doi: 10.3390/biom15020264. Biomolecules. 2025. PMID: 40001567 Free PMC article.
-
Effects of an Adipose Mesenchymal Stem Cell-Derived Conditioned medium and TGF-β1 on Human Keratinocytes In Vitro.Int J Mol Sci. 2023 Sep 29;24(19):14726. doi: 10.3390/ijms241914726. Int J Mol Sci. 2023. PMID: 37834173 Free PMC article.
-
[Effects of hypoxia-pretreated rat adipose-derived mesenchymal stem cells conditioned medium on wound healing of rats with full-thickness defects].Zhonghua Shao Shang Za Zhi. 2020 Sep 20;36(9):803-812. doi: 10.3760/cma.j.cn501120-20200508-00258. Zhonghua Shao Shang Za Zhi. 2020. PMID: 32972065 Chinese.
-
The Effects of Adipose Stem Cell-Conditioned Media on Fibrogenesis of Dermal Fibroblasts Stimulated by Transforming Growth Factor-β1.J Burn Care Res. 2018 Jan 1;39(1):129-140. doi: 10.1097/BCR.0000000000000558. J Burn Care Res. 2018. PMID: 29931303 Free PMC article.
-
[Protective effects of estrogen modified hBMSC on HG-induced injury of vascular endothelial cells].Zhongguo Ying Yong Sheng Li Xue Za Zhi. 2022 Jul;38(4):289-294. doi: 10.12047/j.cjap.6244.2022.054. Zhongguo Ying Yong Sheng Li Xue Za Zhi. 2022. PMID: 36414548 Chinese.
Cited by
-
New Insights on the miRNA Role in Diabetic Tendinopathy: Adipose-Derived Mesenchymal Stem Cell Conditioned Medium as a Potential Innovative Epigenetic-Based Therapy for Tendon Healing.Biomolecules. 2025 Feb 11;15(2):264. doi: 10.3390/biom15020264. Biomolecules. 2025. PMID: 40001567 Free PMC article.
-
Preliminary Report on the Time-Related Effect of a Single Autologous Adipose-Derived Mesenchymal Stem Cells Injection in Hip Osteoarthritis: A Retrospective Observational Study.Adv Orthop. 2025 Jun 16;2025:3424035. doi: 10.1155/aort/3424035. eCollection 2025. Adv Orthop. 2025. PMID: 40552343 Free PMC article.
-
Enhancing Tendon Regeneration: Investigating the Impact of Topography on the Secretome of Adipose-Derived Stem Cells.Adv Sci (Weinh). 2025 May;12(18):e2417447. doi: 10.1002/advs.202417447. Epub 2025 Mar 17. Adv Sci (Weinh). 2025. PMID: 40091553 Free PMC article.
References
-
- Maffulli N, Khan KM, Puddu G. Overuse tendon conditions: time to change a confusing terminology. Arthrosc J Arthrosc Relat Surg 1998; 14: 840–843. - PubMed
-
- Lui PPY. Tendinopathy in diabetes mellitus patients-epidemiology, pathogenesis, and management. Scand J Med Sci Sports 2017; 27: 776–787. - PubMed
-
- D’Amora M, Mondillo F, Cantalupo T, et al.. Achilles tendinopathy and patellar tendon: integrated imaging. Minerva Ortop E Traumatol. Epub ahead of print January 2021. DOI: 10.23736/S0394-3410.20.03984-3. - DOI
-
- Kellam JF, Hunter GA, McElwain JP. Review of the operative treatment of Achilles tendon rupture. Clin Orthop 1985; 201: 80–83. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

