Safety and Tolerability of Intra-Articular Injection of Adipose-Derived Mesenchymal Stem Cells GXCPC1 in 11 Subjects With Knee Osteoarthritis: A Nonrandomized Pilot Study Without a Control Arm
- PMID: 38205679
- PMCID: PMC10785714
- DOI: 10.1177/09636897231221882
Safety and Tolerability of Intra-Articular Injection of Adipose-Derived Mesenchymal Stem Cells GXCPC1 in 11 Subjects With Knee Osteoarthritis: A Nonrandomized Pilot Study Without a Control Arm
Abstract
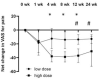
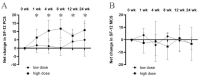
The current study aimed to determine the safety profile of intra-articular-injected allogeneic adipose-derived mesenchymal stem cells (ADSCs) GXCPC1 in subjects with knee osteoarthritis (OA) and its preliminary efficacy outcome. The 3 + 3 phase I study was designed with two dose-escalation cohorts: low dose (6.7 × 106 GXCPC1, N = 5) and high dose (4 × 107 GXCPC1, N = 6). The primary endpoint was safety, which was evaluated by recording adverse events throughout the trial; the secondary endpoints included total, pain, stiffness, and function subscales of the Western Ontario and McMaster Universities Arthritis Index (WOMAC), Visual Analogue Scale (VAS) for pain, and 12-Item Short Form (SF-12) health survey questionnaire. The GXCPC1 treatment was found to be safe after 1 year of follow-up with no treatment-related severe adverse events observed. When compared to baseline, subjects in both the low- and high-dose cohorts demonstrated improving trends in pain and knee function after receiving GXCPC1 treatment. Generally, the net change in pain (95% confidence interval (CI) = -7.773 to -2.561t at 12 weeks compared to baseline) and knee function (95% CI = -24.297 to -10.036t at 12 weeks compared to baseline) was better in subjects receiving high-dose GXCPC1. Although this study included a limited number of subjects without a placebo arm, it showed that the intra-articular injection of ADSCs was safe and well-tolerated in subjects with therapeutic alternatives to treat knee OA. However, a larger scale study with an appropriate control would be necessary for clinical efficacy in the following study.
Trial registration: ClinicalTrials.gov NCT03943576.
Keywords: GXCPC1; allogeneic adipose-derived mesenchymal stem cells (ADSCs); knee osteoarthritis; safety; tolerability.
Conflict of interest statement
Declaration of Conflicting InterestsThe author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Figures

Similar articles
-
Adipose Mesenchymal Stromal Cell-Based Therapy for Severe Osteoarthritis of the Knee: A Phase I Dose-Escalation Trial.Stem Cells Transl Med. 2016 Jul;5(7):847-56. doi: 10.5966/sctm.2015-0245. Epub 2016 May 23. Stem Cells Transl Med. 2016. PMID: 27217345 Free PMC article. Clinical Trial.
-
Clinical Efficacy and Safety of the Intra-articular Injection of Autologous Adipose-Derived Mesenchymal Stem Cells for Knee Osteoarthritis: A Phase III, Randomized, Double-Blind, Placebo-Controlled Trial.Am J Sports Med. 2023 Jul;51(9):2243-2253. doi: 10.1177/03635465231179223. Epub 2023 Jun 21. Am J Sports Med. 2023. PMID: 37345256 Clinical Trial.
-
Intra-articular injections of allogeneic human adipose-derived mesenchymal progenitor cells in patients with symptomatic bilateral knee osteoarthritis: a Phase I pilot study.Regen Med. 2020 May;15(5):1625-1636. doi: 10.2217/rme-2019-0106. Epub 2020 Jul 17. Regen Med. 2020. PMID: 32677876 Clinical Trial.
-
Safety and efficacy of adipose-derived mesenchymal stem cells for knee osteoarthritis: A systematic review and m-analysis.Joint Bone Spine. 2022 Oct;89(5):105404. doi: 10.1016/j.jbspin.2022.105404. Epub 2022 May 6. Joint Bone Spine. 2022. PMID: 35534001
-
Intra-articular Injection of Autologous Adipose-Derived Stem Cells or Stromal Vascular Fractions: Are They Effective for Patients With Knee Osteoarthritis? A Systematic Review With Meta-analysis of Randomized Controlled Trials.Am J Sports Med. 2023 Mar;51(3):837-848. doi: 10.1177/03635465211053893. Epub 2022 Jan 12. Am J Sports Med. 2023. PMID: 35019764
Cited by
-
Unlocking Precision Medicine: Liquid Biopsy Advancements in Renal Cancer Detection and Monitoring.Int J Mol Sci. 2024 Mar 30;25(7):3867. doi: 10.3390/ijms25073867. Int J Mol Sci. 2024. PMID: 38612677 Free PMC article. Review.
-
Therapeutic Potential of Adipose Mesenchymal Stem Cells for Synovial Regeneration: from In-Vitro Studies to Clinical Applications.Stem Cell Rev Rep. 2025 Aug;21(6):1710-1727. doi: 10.1007/s12015-025-10909-5. Epub 2025 Jun 10. Stem Cell Rev Rep. 2025. PMID: 40493163 Free PMC article. Review.
-
Algofunctional outcome after intra-articular bipolar pulsed radiofrequency ablation for pain in osteoarthritis of the knee: A retrospective study.Medicine (Baltimore). 2024 Nov 15;103(46):e40504. doi: 10.1097/MD.0000000000040504. Medicine (Baltimore). 2024. PMID: 39560582 Free PMC article.
-
Advancements in Regenerative Therapies for Orthopedics: A Comprehensive Review of Platelet-Rich Plasma, Mesenchymal Stem Cells, Peptide Therapies, and Biomimetic Applications.J Clin Med. 2025 Mar 18;14(6):2061. doi: 10.3390/jcm14062061. J Clin Med. 2025. PMID: 40142869 Free PMC article. Review.
-
Intra-Articular Injection of Human Bone Marrow-Derived Mesenchymal Stem Cells in Knee Osteoarthritis: A Randomized, Double-Blind, Controlled Trial.Cell Transplant. 2025 Jan-Dec;34:9636897241303275. doi: 10.1177/09636897241303275. Cell Transplant. 2025. PMID: 39874108 Free PMC article. Clinical Trial.
References
-
- Quicke JG, Conaghan PG, Corp N, Peat G. Osteoarthritis year in review 2021: epidemiology & therapy. Osteoarthritis Cartilage. 2022;30(2):196–206. - PubMed
-
- Arden NK, Perry TA, Bannuru RR, Bruyère O, Cooper C, Haugen IK, Hochberg MC, McAlindon TE, Mobasheri A, Reginster JY. Non-surgical management of knee osteoarthritis: comparison of ESCEO and OARSI 2019 guidelines. Nat Rev Rheumatol. 2021;17(1):59–66. - PubMed
-
- Hunter DJ, Bierma-Zeinstra S. Osteoarthritis. Lancet. 2019;393(10182):1745–59. - PubMed
Publication types
MeSH terms
Associated data
LinkOut - more resources
Full Text Sources
Medical

