ANGPTL4 accelerates ovarian serous cystadenocarcinoma carcinogenesis and angiogenesis in the tumor microenvironment by activating the JAK2/STAT3 pathway and interacting with ESM1
- PMID: 38212795
- PMCID: PMC10785435
- DOI: 10.1186/s12967-023-04819-8
ANGPTL4 accelerates ovarian serous cystadenocarcinoma carcinogenesis and angiogenesis in the tumor microenvironment by activating the JAK2/STAT3 pathway and interacting with ESM1
Abstract
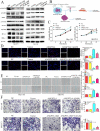
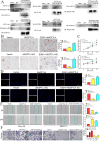
Background: Ovarian cancer (OC) is a malignant neoplasm that displays increased vascularization. Angiopoietin-like 4 (ANGPTL4) is a secreted glycoprotein that functions as a regulator of cell metabolism and angiogenesis and plays a critical role in tumorigenesis. However, the precise role of ANGPTL4 in the OC microenvironment, particularly its involvement in angiogenesis, has not been fully elucidated.
Methods: The expression of ANGPTL4 was confirmed by bioinformatics and IHC in OC. The potential molecular mechanism of ANGPTL4 was measured by RNA-sequence. We used a series of molecular biological experiments to measure the ANGPTL4-JAK2-STAT3 and ANGPTL4-ESM1 axis in OC progression, including MTT, EdU, wound healing, transwell, xenograft model, oil red O staining, chick chorioallantoic membrane assay and zebrafish model. Moreover, the molecular mechanisms were confirmed by Western blot, Co-IP and molecular docking.
Results: Our study demonstrates a significant upregulation of ANGPTL4 in OC specimens and its strong association with unfavorable prognosis. RNA-seq analysis affirms that ANGPTL4 facilitates OC development by driving JAK2-STAT3 signaling pathway activation. The interaction between ANGPTL4 and ESM1 promotes ANGPTL4 binding to lipoprotein lipase (LPL), thereby resulting in reprogrammed lipid metabolism and the promotion of OC cell proliferation, migration, and invasion. In the OC microenvironment, ESM1 may interfere with the binding of ANGPTL4 to integrin and vascular-endothelial cadherin (VE-Cad), which leads to stabilization of vascular integrity and ultimately promotes angiogenesis.
Conclusion: Our findings underscore that ANGPTL4 promotes OC development via JAK signaling and induces angiogenesis in the tumor microenvironment through its interaction with ESM1.
Keywords: ANGPTL4; Angiogenesis; ESM1; JAK2/STAT3 pathway; Ovarian cancer.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures





References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

