Lipoxins A4 and B4 inhibit glial cell activation via CXCR3 signaling in acute retinal neuroinflammation
- PMID: 38212822
- PMCID: PMC10782675
- DOI: 10.1186/s12974-024-03010-0
Lipoxins A4 and B4 inhibit glial cell activation via CXCR3 signaling in acute retinal neuroinflammation
Abstract
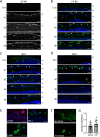
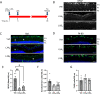
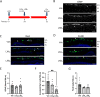
Lipoxins are small lipids that are potent endogenous mediators of systemic inflammation resolution in a variety of diseases. We previously reported that Lipoxins A4 and B4 (LXA4 and LXB4) have protective activities against neurodegenerative injury. Yet, lipoxin activities and downstream signaling in neuroinflammatory processes are not well understood. Here, we utilized a model of posterior uveitis induced by lipopolysaccharide endotoxin (LPS), which results in rapid retinal neuroinflammation primarily characterized by activation of resident macroglia (astrocytes and Müller glia), and microglia. Using this model, we observed that each lipoxin reduces acute inner retinal inflammation by affecting endogenous glial responses in a cascading sequence beginning with astrocytes and then microglia, depending on the timing of exposure; prophylactic or therapeutic. Subsequent analyses of retinal cytokines and chemokines revealed inhibition of both CXCL9 (MIG) and CXCL10 (IP10) by each lipoxin, compared to controls, following LPS injection. CXCL9 and CXCL10 are common ligands for the CXCR3 chemokine receptor, which is prominently expressed in inner retinal astrocytes and ganglion cells. We found that CXCR3 inhibition reduces LPS-induced neuroinflammation, while CXCR3 agonism alone induces astrocyte reactivity. Together, these data uncover a novel lipoxin-CXCR3 pathway to promote distinct anti-inflammatory and proresolution cascades in endogenous retinal glia.
Keywords: CXCL10; CXCL9; CXCR3; Gliosis; Inflammation resolution; Lipoxins; Neuroinflammation; Retina; Uveitis.
© 2024. The Author(s).
Conflict of interest statement
Several authors (I. L-B, J.M.S., K.G.) have obtained a US patent for use of lipoxins to treat neurodegeneration.
Figures


References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

