15NRORC: An Azine Labeling Protocol
- PMID: 38215470
- PMCID: PMC12012823
- DOI: 10.1021/jacs.3c11618
15NRORC: An Azine Labeling Protocol
Abstract
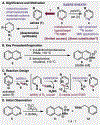
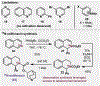
A practical method for the synthesis of 15N-labeled azines with a high degree of isotopic enrichment is described. Activation of azine heterocycles with an electron-deficient arene allows for the facile substitution of the nitrogen atom with a specifically designed 15N-labeled reagent that undergoes a canonical ANRORC-type mechanism. A wide range of azines can be converted to their corresponding 15N isotopologs using this method, and it also allows for dearomative access to reduced heterocyclic congeners. A short dearomative formal synthesis of 15N-solifenacin is accomplished as well to demonstrate a practical application of this method for generating labeled pharmaceuticals.
Conflict of interest statement
The authors declare no competing financial interest.
Figures

References
-
- Vitaku E; Smith DT; Njardarson JT Analysis of the Structural Diversity, Substitution Patterns, and Frequency of Nitrogen Heterocycles among U.S. FDA Approved Pharmaceuticals. J. Med. Chem 2014, 57 (24), 10257–10274. - PubMed
-
- Theillet F-X In-Cell Structural Biology by NMR: The Benefits of the Atomic Scale. Chem. Rev 2022, 122 (10), 9497–9570. - PubMed
-
- Nakabayashi R; Mori T; Takeda N; Toyooka K; Sudo H; Tsugawa H; Saito K Metabolomics with 15N Labeling for Characterizing Missing Monoterpene Indole Alkaloids in Plants. Anal. Chem 2020, 92 (8), 5670–5675. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

