KLF5 inhibits the migration and invasion in cervical cancer cell lines by regulating SNAI1
- PMID: 38217587
- PMCID: PMC11191462
- DOI: 10.3233/CBM-230175
KLF5 inhibits the migration and invasion in cervical cancer cell lines by regulating SNAI1
Abstract
Background: Epithelial-mesenchymal transition (EMT) is an important biological process by which malignant tumor cells to acquire migration and invasion abilities. This study explored the role of KLF5 in the EMT process of in cervical cancer cell lines.
Objective: Krüpple-like factor 5 (KLF5) is a basic transcriptional factor that plays a key role in cell-cycle arrest and inhibition of apoptosis. However, the molecular mechanism by which KLF5 mediates the biological functions of cervical cancer cell lines has not been elucidated. Here, we focus on the potential function of ELF5 in regulating the EMT process in in vitro model of cervical cancer cell lines.
Method: Western-blot and real-time quantitative PCR were used to detect the expression of EMT-related genes in HeLa cells. MTT assays, cell scratch and Transwell assays were used to assess HeLa cells proliferation and invasion capability. Using the bioinformatics tool JASPAR, we identified a high-scoring KLF5-like binding sequence in the SNAI1 gene promoter. Luciferase reporter assays was used to detect transcriptional activity for different SNAI1 promoter truncates.
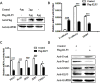
Result: After overexpressing the KLF5 gene in HeLa cells, KLF5 not only significantly inhibited the invasion and migration of HeLa cells, but also increased the expression of E-cadherin and decreased the expression of N-cadherin and MMP9. In addition, the mRNA expression of upstream regulators of E-cadherin, such as SNAI1, SLUG, ZEB1/2 and TWIST1 was also decreased. Furthermore, KLF5 inhibiting the expression of the SNAI1 gene via binding its promoter region, and the EMT of Hela cells was promoted after overexpression of the SNAI1 gene.
Conclusion: These results indicate that KLF5 can downregulate the EMT process of HeLa cells by decreasing the expression of the SNAI1 gene, thereby inhibiting the migration and invasion of HeLa cervical cancer cells.
Keywords: EMT; KLF5; SNAI1; cervical cancer cells.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures






Similar articles
-
KLF5 silence attenuates proliferation and epithelial-mesenchymal transition induction in Hep-2 cells through NF-κB signaling pathway.Eur Rev Med Pharmacol Sci. 2019 May;23(9):3867-3875. doi: 10.26355/eurrev_201905_17814. Eur Rev Med Pharmacol Sci. 2019. PMID: 31115014
-
FoxM1 overexpression promotes epithelial-mesenchymal transition and metastasis of hepatocellular carcinoma.World J Gastroenterol. 2015 Jan 7;21(1):196-213. doi: 10.3748/wjg.v21.i1.196. World J Gastroenterol. 2015. PMID: 25574092 Free PMC article.
-
Novel ESCC-related gene ZNF750 as potential Prognostic biomarker and inhibits Epithelial-Mesenchymal Transition through directly depressing SNAI1 promoter in ESCC.Theranostics. 2020 Jan 1;10(4):1798-1813. doi: 10.7150/thno.38210. eCollection 2020. Theranostics. 2020. PMID: 32042337 Free PMC article.
-
ML264: An Antitumor Agent that Potently and Selectively Inhibits Krüppel-like Factor Five (KLF5) Expression: A Probe for Studying Colon Cancer Development and Progression.2011 Oct 31 [updated 2013 Mar 7]. In: Probe Reports from the NIH Molecular Libraries Program [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2010–. 2011 Oct 31 [updated 2013 Mar 7]. In: Probe Reports from the NIH Molecular Libraries Program [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2010–. PMID: 23762940 Free Books & Documents. Review.
-
Anti-metastatic potential of flavonoids for the treatment of cancers: focus on epithelial-mesenchymal transition (EMT) process.Naunyn Schmiedebergs Arch Pharmacol. 2025 May 28. doi: 10.1007/s00210-025-04235-3. Online ahead of print. Naunyn Schmiedebergs Arch Pharmacol. 2025. PMID: 40434422 Review.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

