Wharton's Jelly mesenchymal stromal cell-derived extracellular vesicles promote nucleus pulposus cell anabolism in an in vitro 3D alginate-bead culture model
- PMID: 38222813
- PMCID: PMC10782051
- DOI: 10.1002/jsp2.1274
Wharton's Jelly mesenchymal stromal cell-derived extracellular vesicles promote nucleus pulposus cell anabolism in an in vitro 3D alginate-bead culture model
Abstract
Background: Intradiscal transplantation of mesenchymal stromal cells (MSCs) has emerged as a promising therapy for intervertebral disc degeneration (IDD). However, the hostile microenvironment of the intervertebral disc (IVD) may compromise the survival of implanted cells. Interestingly, studies reported that paracrine factors, such as extracellular vesicles (EVs) released by MSCs, may regenerate the IVD. The aim of this study was to investigate the therapeutic effects of Wharton's Jelly MSC (WJ-MSC)-derived EVs on human nucleus pulposus cells (hNPCs) using an in vitro 3D alginate-bead culture model.
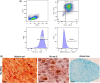
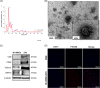
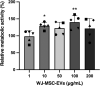
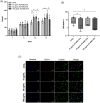
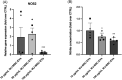
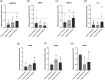
Methods: After EV isolation and characterization, hNPCs isolated from surgical specimens were encapsulated in alginate beads and treated with 10, 50, and 100 μg/mL WJ-MSC-EVs. Cell proliferation and viability were assessed by flow cytometry and live/dead staining. Nitrite and glycosaminoglycan (GAG) content was evaluated through Griess and 1,9-dimethylmethylene blue assays. hNPCs in alginate beads were paraffin-embedded and stained for histological analysis (hematoxylin-eosin and Alcian blue) to assess extracellular matrix (ECM) composition. Gene expression levels of catabolic (MMP1, MMP13, ADAMTS5, IL6, NOS2), anabolic (ACAN), and hNPC marker (SOX9, KRT19) genes were analyzed through qPCR. Collagen type I and type II content was assessed with Western blot analysis.
Results: Treatment with WJ-MSC-EVs resulted in an increase in cell content and a decrease in cell death in degenerated hNPCs. Nitrite production was drastically reduced by EV treatment compared to the control. Furthermore, proteoglycan content was enhanced and confirmed by Alcian blue histological staining. EV stimulation attenuated ECM degradation and inflammation by suppressing catabolic and inflammatory gene expression levels. Additionally, NPC phenotypic marker genes were also maintained by the EV treatment.
Conclusions: WJ-MSC-derived EVs ameliorated hNPC growth and viability, and attenuated ECM degradation and oxidative stress, offering new opportunities for IVD regeneration as an attractive alternative strategy to cell therapy, which may be jeopardized by the harsh microenvironment of the IVD.
Keywords: exosome; extracellular vesicles; intervertebral disc; intervertebral disc degeneration; intervertebral disc regeneration; low back pain; mesenchymal stromal cells.
© 2023 The Authors. JOR Spine published by Wiley Periodicals LLC on behalf of Orthopaedic Research Society.
Conflict of interest statement
GV is an Editorial Board member of JOR Spine and co‐author of this article. He was excluded from editorial decision‐making related to the acceptance of this article for publication in the journal.
Figures


References
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

