Robust explanation supervision for false positive reduction in pulmonary nodule detection
- PMID: 38224306
- PMCID: PMC10939846
- DOI: 10.1002/mp.16937
Robust explanation supervision for false positive reduction in pulmonary nodule detection
Abstract
Background: Lung cancer is the deadliest and second most common cancer in the United States due to the lack of symptoms for early diagnosis. Pulmonary nodules are small abnormal regions that can be potentially correlated to the occurrence of lung cancer. Early detection of these nodules is critical because it can significantly improve the patient's survival rates. Thoracic thin-sliced computed tomography (CT) scanning has emerged as a widely used method for diagnosing and prognosis lung abnormalities.
Purpose: The standard clinical workflow of detecting pulmonary nodules relies on radiologists to analyze CT images to assess the risk factors of cancerous nodules. However, this approach can be error-prone due to the various nodule formation causes, such as pollutants and infections. Deep learning (DL) algorithms have recently demonstrated remarkable success in medical image classification and segmentation. As an ever more important assistant to radiologists in nodule detection, it is imperative ensure the DL algorithm and radiologist to better understand the decisions from each other. This study aims to develop a framework integrating explainable AI methods to achieve accurate pulmonary nodule detection.
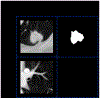
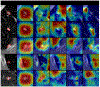
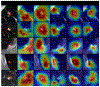
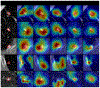
Methods: A robust and explainable detection (RXD) framework is proposed, focusing on reducing false positives in pulmonary nodule detection. Its implementation is based on an explanation supervision method, which uses nodule contours of radiologists as supervision signals to force the model to learn nodule morphologies, enabling improved learning ability on small dataset, and enable small dataset learning ability. In addition, two imputation methods are applied to the nodule region annotations to reduce the noise within human annotations and allow the model to have robust attributions that meet human expectations. The 480, 265, and 265 CT image sets from the public Lung Image Database Consortium and Image Database Resource Initiative (LIDC-IDRI) dataset are used for training, validation, and testing.
Results: Using only 10, 30, 50, and 100 training samples sequentially, our method constantly improves the classification performance and explanation quality of baseline in terms of Area Under the Curve (AUC) and Intersection over Union (IoU). In particular, our framework with a learnable imputation kernel improves IoU from baseline by 24.0% to 80.0%. A pre-defined Gaussian imputation kernel achieves an even greater improvement, from 38.4% to 118.8% from baseline. Compared to the baseline trained on 100 samples, our method shows less drop in AUC when trained on fewer samples. A comprehensive comparison of interpretability shows that our method aligns better with expert opinions.
Conclusions: A pulmonary nodule detection framework was demonstrated using public thoracic CT image datasets. The framework integrates the robust explanation supervision (RES) technique to ensure the performance of nodule classification and morphology. The method can reduce the workload of radiologists and enable them to focus on the diagnosis and prognosis of the potential cancerous pulmonary nodules at the early stage to improve the outcomes for lung cancer patients.
Keywords: deep learning; explainable AI; pulmonary nodule detection.
© 2024 American Association of Physicists in Medicine.
Conflict of interest statement
Conflict of interest
The authors have no conflict of interests to disclose.
Figures







Similar articles
-
An adaptive morphology based segmentation technique for lung nodule detection in thoracic CT image.Comput Methods Programs Biomed. 2020 Dec;197:105720. doi: 10.1016/j.cmpb.2020.105720. Epub 2020 Aug 25. Comput Methods Programs Biomed. 2020. PMID: 32877818
-
Computer-aided detection of pulmonary nodules: a comparative study using the public LIDC/IDRI database.Eur Radiol. 2016 Jul;26(7):2139-47. doi: 10.1007/s00330-015-4030-7. Epub 2015 Oct 6. Eur Radiol. 2016. PMID: 26443601 Free PMC article.
-
Computer-Aided Diagnosis (CAD) of Pulmonary Nodule of Thoracic CT Image Using Transfer Learning.J Digit Imaging. 2019 Dec;32(6):995-1007. doi: 10.1007/s10278-019-00204-4. J Digit Imaging. 2019. PMID: 31044393 Free PMC article.
-
A survey of computer-aided diagnosis of lung nodules from CT scans using deep learning.Comput Biol Med. 2021 Oct;137:104806. doi: 10.1016/j.compbiomed.2021.104806. Epub 2021 Aug 25. Comput Biol Med. 2021. PMID: 34461501 Review.
-
[Research progress on computed tomography image detection and classification of pulmonary nodule based on deep learning].Sheng Wu Yi Xue Gong Cheng Xue Za Zhi. 2019 Aug 25;36(4):670-676. doi: 10.7507/1001-5515.201806019. Sheng Wu Yi Xue Gong Cheng Xue Za Zhi. 2019. PMID: 31441270 Free PMC article. Review. Chinese.
Cited by
-
Diagnostic performance and safety for robotic-assisted bronchoscopy in pulmonary nodules: a systematic review and meta-analysis.Int J Surg. 2025 Jun 1;111(6):4020-4032. doi: 10.1097/JS9.0000000000002423. Epub 2025 May 12. Int J Surg. 2025. PMID: 40358662 Free PMC article.
References
-
- Investigators IELCAP. Survival of patients with stage I lung cancer detected on CT screening. New England Journal of Medicine. 2006;355(17):1763–1771. - PubMed
-
- Hu M, Wang J, Chang C-W, Liu T, Yang X. End-to-end brain tumor detection using a graph-feature-based classifier. Vol 12468: SPIE; 2023.
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

