EphrinB2 knockdown in cervical spinal cord preserves diaphragm innervation in a mutant SOD1 mouse model of ALS
- PMID: 38224498
- PMCID: PMC10945582
- DOI: 10.7554/eLife.89298
EphrinB2 knockdown in cervical spinal cord preserves diaphragm innervation in a mutant SOD1 mouse model of ALS
Abstract
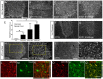
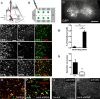
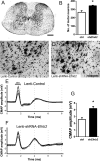
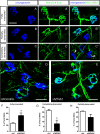
Amyotrophic lateral sclerosis (ALS) is a neurodegenerative disease characterized by motor neuron loss. Importantly, non-neuronal cell types such as astrocytes also play significant roles in disease pathogenesis. However, mechanisms of astrocyte contribution to ALS remain incompletely understood. Astrocyte involvement suggests that transcellular signaling may play a role in disease. We examined contribution of transmembrane signaling molecule ephrinB2 to ALS pathogenesis, in particular its role in driving motor neuron damage by spinal cord astrocytes. In symptomatic SOD1G93A mice (a well-established ALS model), ephrinB2 expression was dramatically increased in ventral horn astrocytes. Reducing ephrinB2 in the cervical spinal cord ventral horn via viral-mediated shRNA delivery reduced motor neuron loss and preserved respiratory function by maintaining phrenic motor neuron innervation of diaphragm. EphrinB2 expression was also elevated in human ALS spinal cord. These findings implicate ephrinB2 upregulation as both a transcellular signaling mechanism in mutant SOD1-associated ALS and a promising therapeutic target.
Keywords: ALS; astrocyte; ephrin; human; motor neuron; mouse; neuroscience; respiratory.
© 2023, Urban et al.
Conflict of interest statement
MU, BC, NH, SM, LS, WZ, EB, NH, ST, BG, RC, DT, PP, MW, MD, AL No competing interests declared
Figures

Update of
-
EphrinB2 knockdown in cervical spinal cord preserves diaphragm innervation in a mutant SOD1 mouse model of ALS.bioRxiv [Preprint]. 2023 Oct 30:2023.05.10.538887. doi: 10.1101/2023.05.10.538887. bioRxiv. 2023. Update in: Elife. 2024 Jan 15;12:RP89298. doi: 10.7554/eLife.89298. PMID: 37215009 Free PMC article. Updated. Preprint.
Similar articles
-
EphrinB2 knockdown in cervical spinal cord preserves diaphragm innervation in a mutant SOD1 mouse model of ALS.bioRxiv [Preprint]. 2023 Oct 30:2023.05.10.538887. doi: 10.1101/2023.05.10.538887. bioRxiv. 2023. Update in: Elife. 2024 Jan 15;12:RP89298. doi: 10.7554/eLife.89298. PMID: 37215009 Free PMC article. Updated. Preprint.
-
GLT1 overexpression in SOD1(G93A) mouse cervical spinal cord does not preserve diaphragm function or extend disease.Neurobiol Dis. 2015 Jun;78:12-23. doi: 10.1016/j.nbd.2015.03.010. Epub 2015 Mar 25. Neurobiol Dis. 2015. PMID: 25818008
-
Microglia RAGE exacerbates the progression of neurodegeneration within the SOD1G93A murine model of amyotrophic lateral sclerosis in a sex-dependent manner.J Neuroinflammation. 2021 Jun 15;18(1):139. doi: 10.1186/s12974-021-02191-2. J Neuroinflammation. 2021. PMID: 34130712 Free PMC article.
-
Astrocytes in Amyotrophic Lateral Sclerosis.In: Araki T, editor. Amyotrophic Lateral Sclerosis [Internet]. Brisbane (AU): Exon Publications; 2021 Jul 25. Chapter 3. In: Araki T, editor. Amyotrophic Lateral Sclerosis [Internet]. Brisbane (AU): Exon Publications; 2021 Jul 25. Chapter 3. PMID: 34473439 Free Books & Documents. Review.
-
Therapeutic Potential of Astrocyte Transplantation.Cell Transplant. 2022 Jan-Dec;31:9636897221105499. doi: 10.1177/09636897221105499. Cell Transplant. 2022. PMID: 35770772 Free PMC article. Review.
Cited by
-
The Diverse Roles of Reactive Astrocytes in the Pathogenesis of Amyotrophic Lateral Sclerosis.Brain Sci. 2024 Feb 4;14(2):158. doi: 10.3390/brainsci14020158. Brain Sci. 2024. PMID: 38391732 Free PMC article. Review.
-
Activation of EphrinB2/EphB2 signaling in the spine cord alters glia-neuron interactions in mice with visceral hyperalgesia following maternal separation.Front Pharmacol. 2024 Sep 3;15:1463339. doi: 10.3389/fphar.2024.1463339. eCollection 2024. Front Pharmacol. 2024. PMID: 39290870 Free PMC article.
References
-
- Alkaslasi MR, Piccus ZE, Hareendran S, Silberberg H, Chen L, Zhang Y, Petros TJ, Le Pichon CE. Single nucleus RNA-sequencing defines unexpected diversity of cholinergic neuron types in the adult mouse spinal cord. Nature Communications. 2021;12:2471. doi: 10.1038/s41467-021-22691-2. - DOI - PMC - PubMed
-
- Andersen PM, Nilsson P, Keränen ML, Forsgren L, Hägglund J, Karlsborg M, Ronnevi LO, Gredal O, Marklund SL. Phenotypic heterogeneity in motor neuron disease patients with CuZn-superoxide dismutase mutations in Scandinavia. Brain. 1997;120 ( Pt 10):1723–1737. doi: 10.1093/brain/120.10.1723. - DOI - PubMed
-
- Blum JA, Klemm S, Shadrach JL, Guttenplan KA, Nakayama L, Kathiria A, Hoang PT, Gautier O, Kaltschmidt JA, Greenleaf WJ, Gitler AD. Single-cell transcriptomic analysis of the adult mouse spinal cord reveals molecular diversity of autonomic and skeletal motor neurons. Nature Neuroscience. 2021;24:572–583. doi: 10.1038/s41593-020-00795-0. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

