Transplantation of human placental chorionic plate-derived mesenchymal stem cells for repair of neurological damage in neonatal hypoxic-ischemic encephalopathy
- PMID: 38227532
- PMCID: PMC11040304
- DOI: 10.4103/1673-5374.390952
Transplantation of human placental chorionic plate-derived mesenchymal stem cells for repair of neurological damage in neonatal hypoxic-ischemic encephalopathy
Abstract
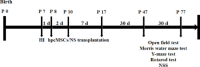
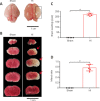
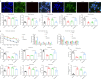
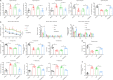
JOURNAL/nrgr/04.03/01300535-202409000-00035/figure1/v/2024-01-16T170235Z/r/image-tiff Neonatal hypoxic-ischemic encephalopathy is often associated with permanent cerebral palsy, neurosensory impairments, and cognitive deficits, and there is no effective treatment for complications related to hypoxic-ischemic encephalopathy. The therapeutic potential of human placental chorionic plate-derived mesenchymal stem cells for various diseases has been explored. However, the potential use of human placental chorionic plate-derived mesenchymal stem cells for the treatment of neonatal hypoxic-ischemic encephalopathy has not yet been investigated. In this study, we injected human placental chorionic plate-derived mesenchymal stem cells into the lateral ventricle of a neonatal hypoxic-ischemic encephalopathy rat model and observed significant improvements in both cognitive and motor function. Protein chip analysis showed that interleukin-3 expression was significantly elevated in neonatal hypoxic-ischemic encephalopathy model rats. Following transplantation of human placental chorionic plate-derived mesenchymal stem cells, interleukin-3 expression was downregulated. To further investigate the role of interleukin-3 in neonatal hypoxic-ischemic encephalopathy, we established an in vitro SH-SY5Y cell model of hypoxic-ischemic injury through oxygen-glucose deprivation and silenced interleukin-3 expression using small interfering RNA. We found that the activity and proliferation of SH-SY5Y cells subjected to oxygen-glucose deprivation were further suppressed by interleukin-3 knockdown. Furthermore, interleukin-3 knockout exacerbated neuronal damage and cognitive and motor function impairment in rat models of hypoxic-ischemic encephalopathy. The findings suggest that transplantation of hpcMSCs ameliorated behavioral impairments in a rat model of hypoxic-ischemic encephalopathy, and this effect was mediated by interleukin-3-dependent neurological function.
Copyright © 2024 Copyright: © 2024 Neural Regeneration Research.
Conflict of interest statement
Figures



Similar articles
-
hUC-MSCs Exert a Neuroprotective Effect via Anti-apoptotic Mechanisms in a Neonatal HIE Rat Model.Cell Transplant. 2019 Dec;28(12):1552-1559. doi: 10.1177/0963689719874769. Epub 2019 Sep 12. Cell Transplant. 2019. PMID: 31512502 Free PMC article.
-
Therapeutic effect of human umbilical cord mesenchymal stem cells on neonatal rat hypoxic-ischemic encephalopathy.J Neurosci Res. 2014 Jan;92(1):35-45. doi: 10.1002/jnr.23304. Epub 2013 Oct 28. J Neurosci Res. 2014. PMID: 24265136
-
Scutellarin ameliorates neonatal hypoxic-ischemic encephalopathy associated with GAP43-dependent signaling pathway.Chin Med. 2021 Oct 18;16(1):105. doi: 10.1186/s13020-021-00517-z. Chin Med. 2021. PMID: 34663387 Free PMC article.
-
Cell-based treatment for perinatal hypoxic-ischemic encephalopathy.Brain Circ. 2021 Mar 30;7(1):13-17. doi: 10.4103/bc.bc_7_21. eCollection 2021 Jan-Mar. Brain Circ. 2021. PMID: 34084971 Free PMC article. Review.
-
Progress in the treatment of neonatal hypoxic-ischemic encephalopathy with umbilical cord blood mononuclear cells.Brain Dev. 2023 Nov;45(10):533-546. doi: 10.1016/j.braindev.2023.09.001. Epub 2023 Oct 6. Brain Dev. 2023. PMID: 37806836 Review.
Cited by
-
Mesenchymal stem cells for peripheral nerve injury and regeneration: a bibliometric and visualization study.Front Neurol. 2024 Aug 5;15:1420402. doi: 10.3389/fneur.2024.1420402. eCollection 2024. Front Neurol. 2024. PMID: 39161869 Free PMC article.
References
-
- Antoniadou E, David AL. Placental stem cells. Best Pract Res Clin Obstet Gynaecol. 2016;31:13–29. - PubMed
-
- Bailo M, Soncini M, Vertua E, Signoroni PB, Sanzone S, Lombardi G, Arienti D, Calamani F, Zatti D, Paul P, Albertini A, Zorzi F, Cavagnini A, Candotti F, Wengler GS, Parolini O. Engraftment potential of human amnion and chorion cells derived from term placenta. Transplantation. 2004;78:1439–1448. - PubMed
LinkOut - more resources
Full Text Sources

