Tumor resistance to anti-mesothelin CAR-T cells caused by binding to shed mesothelin is overcome by targeting a juxtamembrane epitope
- PMID: 38227666
- PMCID: PMC10823246
- DOI: 10.1073/pnas.2317283121
Tumor resistance to anti-mesothelin CAR-T cells caused by binding to shed mesothelin is overcome by targeting a juxtamembrane epitope
Abstract
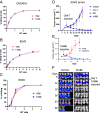
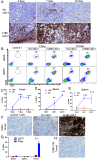
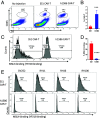
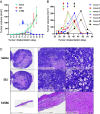
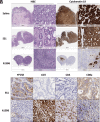
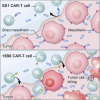
Despite many clinical trials, CAR-T cells are not yet approved for human solid tumor therapy. One popular target is mesothelin (MSLN) which is highly expressed on the surface of about 30% of cancers including mesothelioma and cancers of the ovary, pancreas, and lung. MSLN is shed by proteases that cleave near the C terminus, leaving a short peptide attached to the cell. Most anti-MSLN antibodies bind to shed MSLN, which can prevent their binding to target cells. To overcome this limitation, we developed an antibody (15B6) that binds next to the membrane at the protease-sensitive region, does not bind to shed MSLN, and makes CAR-T cells that have much higher anti-tumor activity than a CAR-T that binds to shed MSLN. We have now humanized the Fv (h15B6), so the CAR-T can be used to treat patients and show that h15B6 CAR-T produces complete regressions in a hard-to-treat pancreatic cancer patient derived xenograft model, whereas CAR-T targeting a shed epitope (SS1) have no anti-tumor activity. In these pancreatic cancers, the h15B6 CAR-T replicates and replaces the cancer cells, whereas there are no CAR-T cells in the tumors receiving SS1 CAR-T. To determine the mechanism accounting for high activity, we used an OVCAR-8 intraperitoneal model to show that poorly active SS1-CAR-T cells are bound to shed MSLN, whereas highly active h15B6 CAR-T do not contain bound MSLN enabling them to bind to and kill cancer cells.
Keywords: antibody; cancer; immunotherapy; ovarian cancer; pancreatic cancer.
Conflict of interest statement
Competing interests statement:X.F.L., M.O., M.H., and I.P. are inventors on patents on 15B6 antibody and have assigned all rights to NIH. One of the reviewers (M.G.R.) of this paper is working with a company that has licensed an anti-MSLN antibody (designated SD1) from NIH that was discovered by NIH employee M.H., a co-author of this paper.
Figures


Similar articles
-
A Bispecific Antibody That Targets the Membrane-Proximal Region of Mesothelin and Retains High Anticancer Activity in the Presence of Shed Mesothelin.Mol Cancer Ther. 2024 Jul 1;23(7):1021–1030. doi: 10.1158/1535-7163.MCT-23-0233. Epub 2024 Apr 22. Mol Cancer Ther. 2024. PMID: 38647528 Free PMC article.
-
Mesothelin antigen density influences anti-mesothelin chimeric antigen receptor T cell cytotoxicity.Cytotherapy. 2024 Apr;26(4):325-333. doi: 10.1016/j.jcyt.2024.01.011. Epub 2024 Feb 13. Cytotherapy. 2024. PMID: 38349311
-
Highly active CAR T cells that bind to a juxtamembrane region of mesothelin and are not blocked by shed mesothelin.Proc Natl Acad Sci U S A. 2022 May 10;119(19):e2202439119. doi: 10.1073/pnas.2202439119. Epub 2022 May 5. Proc Natl Acad Sci U S A. 2022. PMID: 35512094 Free PMC article.
-
Mesothelin-targeted CAR-T cell therapy for solid tumors.Expert Opin Biol Ther. 2021 Apr;21(4):473-486. doi: 10.1080/14712598.2021.1843628. Epub 2020 Nov 12. Expert Opin Biol Ther. 2021. PMID: 33176519 Review.
-
Immunotherapy targeting mesothelin in acute myeloid leukemia.J Leukoc Biol. 2022 Oct;112(4):813-821. doi: 10.1002/JLB.5MR0622-483R. Epub 2022 Aug 10. J Leukoc Biol. 2022. PMID: 35946307 Review.
Cited by
-
ImmunoPET for mesothelin positive tissues using bio-orthogonal in-vivo click chemistry.Nucl Med Biol. 2025 Jul 15;148-149:109051. doi: 10.1016/j.nucmedbio.2025.109051. Online ahead of print. Nucl Med Biol. 2025. PMID: 40694891
-
Mesothelin as a Signal Pathways and Epigenetic Target in Cancer Therapy.Cancers (Basel). 2025 Mar 26;17(7):1118. doi: 10.3390/cancers17071118. Cancers (Basel). 2025. PMID: 40227616 Free PMC article. Review.
-
Repurposing anti-mesothelin CAR-NK immunotherapy against colorectal cancer.J Transl Med. 2024 Dec 4;22(1):1100. doi: 10.1186/s12967-024-05851-y. J Transl Med. 2024. PMID: 39627822 Free PMC article.
-
Immuno-oncology recapitulates ontogeny: Modern cell and gene therapy for cancer.Mol Ther. 2025 May 7;33(5):2229-2237. doi: 10.1016/j.ymthe.2025.03.042. Epub 2025 Mar 27. Mol Ther. 2025. PMID: 40156188 Review.
-
Harnessing γδ T Cells against Human Gynecologic Cancers.Life (Basel). 2024 Feb 29;14(3):325. doi: 10.3390/life14030325. Life (Basel). 2024. PMID: 38541651 Free PMC article. Review.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

