Machine Learning-assisted immunophenotyping of peripheral blood identifies innate immune cells as best predictor of response to induction chemo-immunotherapy in head and neck squamous cell carcinoma - knowledge obtained from the CheckRad-CD8 trial
- PMID: 38232493
- PMCID: PMC10827535
- DOI: 10.1016/j.neo.2023.100953
Machine Learning-assisted immunophenotyping of peripheral blood identifies innate immune cells as best predictor of response to induction chemo-immunotherapy in head and neck squamous cell carcinoma - knowledge obtained from the CheckRad-CD8 trial
Abstract
Purpose: Individual prediction of treatment response is crucial for personalized treatment in multimodal approaches against head-and-neck squamous cell carcinoma (HNSCC). So far, no reliable predictive parameters for treatment schemes containing immunotherapy have been identified. This study aims to predict treatment response to induction chemo-immunotherapy based on the peripheral blood immune status in patients with locally advanced HNSCC.
Methods: The peripheral blood immune phenotype was assessed in whole blood samples in patients treated in the phase II CheckRad-CD8 trial as part of the pre-planned translational research program. Blood samples were analyzed by multicolor flow cytometry before (T1) and after (T2) induction chemo-immunotherapy with cisplatin/docetaxel/durvalumab/tremelimumab. Machine Learning techniques were used to predict pathological complete response (pCR) after induction therapy.
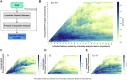
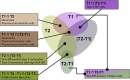
Results: The tested classifier methods (LDA, SVM, LR, RF, DT, and XGBoost) allowed a distinct prediction of pCR. Highest accuracy was achieved with a low number of features represented as principal components. Immune parameters obtained from the absolute difference (lT2-T1l) allowed the best prediction of pCR. In general, less than 30 parameters and at most 10 principal components were needed for highly accurate predictions. Across several datasets, cells of the innate immune system such as polymorphonuclear cells, monocytes, and plasmacytoid dendritic cells are most prominent.
Conclusions: Our analyses imply that alterations of the innate immune cell distribution in the peripheral blood following induction chemo-immuno-therapy is highly predictive for pCR in HNSCC.
Keywords: Chemotherapy; HNSCC; Immune phenotyping; Immunotherapy; Induction therapy.
Copyright © 2024. Published by Elsevier Inc.
Conflict of interest statement
Declaration of competing interest M.H. conflict of interest with Merck Serono (advisory role, speakers’ bureau, honoraria, travel expenses, research funding); MSD (advisory role, speakers’ bureau, honoraria, travel expenses, research funding); AstraZeneca (advisory role, speakers’ bureau, honoraria, travel expenses, research funding); Novartis (research funding); BMS (advisory role, speakers’ bureau, honoraria, travel expenses, research funding); Teva (travel expenses); Sanofi (advisory role, honoraria). M.E. conflict of interest with Diaceutics (employment, honoraria, advisory role, speakers’ bureau, travel expenses); Cepheid (research funding, advisory role); AstraZeneca (honoraria, advisory role, speakers’ bureau, travel expenses); Roche (honoraria, travel expenses); MSD (honoraria, speakers’ bureau); GenomicHealth (honoraria, advisory role, speakers bureau, travel expenses); Astellas (honoraria, speakers’ bureau); Janssen-Cilag (honoraria, advisory role, research funding, travel expenses); Stratifyer (research funding, patents). G.K. conflict of interest with BMS (advisory role); Lilly (advisory role); Roche (advisory role) S.L. conflict of interest with AstraZeneca (honoraria, advisory role); BMS (honoraria, advisory role, speakers’ bureau); MSD (honoraria, advisory role); Merck Serono (honoraria, speakers’ bureau); ISA-Pharmaceuticals (research funding) M.G.H. conflict of interest with Roche (stock); Varian (stock); Sanofi (stock); AstraZeneca (honoraria); BMS (honoraria, advisory role); MSD (honoraria, advisory role); Merck Serono (honoraria); Celgene (honoraria). B.T. conflict of interest with BMS (advisory role, honoraria); Merck Serono (advisory role, speakers’ bureau, honoraria); MSD (advisory role, speakers’ bureau, honoraria); Sanofi (advisory role, honoraria). A.Hi. conflict of interest with Roche (honoraria). A.H. conflict of interest with BMS (honoraria, advisory role); MSD (honoraria, advisory role); Roche (honoraria, advisory role, research funding); AstraZeneca (honoraria, advisory role, research funding); Boehringer Ingelheim (honoraria); Abbvie (honoraria); Cepheid (advisory role, research funding); Quiagen (advisory role); Janssen-Cilag (honoraria, advisory role, research funding); Ipsen (honoraria, advisory role); NanoString Technologies (advisory role, research funding, expert testimony); Illumina (advisory role); 3DHistech (advisory role); Diaceutics (advisory role); BioNTech (research funding). W.B. conflict of interest with BMS (advisory role); MSD (advisory role); Merck Serono (advisory role); Pfitzer (advisory role); AstraZeneca (advisory role). U.S.G. conflict of interest with AstraZeneca (advisory role, research funding); BMS (advisory role); MSD (research funding); MedUpdate (literature research and presentation activities), Dr. Sennewald Medizintechnik (travel expenses and advisory role), Merck (presentation activities). R.F. conflict of interest with MSD (honoraria, advisory role, research funding, travel expenses); Fresenius (honoraria); BrainLab (honoraria); AstraZeneca (honoraria, advisory role, research funding, travel expenses); Merck Serono (advisory role, research funding, travel expenses); Novocure (advisory role, speakers’ bureau, research funding); Sennewald (speakers’ bureau, travel expenses). The other authors declare no conflicts of interest. All other not listed authors do not have a conflict of Interest
Figures

References
-
- Burtness B., Harrington K.J., Greil R., Soulieres D., Tahara M., de Castro G., Jr., Psyrri A., Baste N., Neupane P., Bratland A., et al. Pembrolizumab alone or with chemotherapy versus cetuximab with chemotherapy for recurrent or metastatic squamous cell carcinoma of the head and neck (KEYNOTE-048): a randomised, open-label, phase 3 study. Lancet. 2019;394:1915–1928. doi: 10.1016/S0140-6736(19)32591-7. - DOI - PubMed
-
- Fietkau R., Hecht M., Hofner B., Lubgan D., Iro H., Gefeller O., Rodel C., Hautmann M.G., Kolbl O., Salay A., et al. Randomized phase-III-trial of concurrent chemoradiation for locally advanced head and neck cancer comparing dose reduced radiotherapy with paclitaxel/cisplatin to standard radiotherapy with fluorouracil/cisplatin: The PacCis-trial. Radiother. Oncol. 2020;144:209–217. doi: 10.1016/j.radonc.2020.01.016. - DOI - PubMed
-
- Blanchard P., Bourhis J., Lacas B., Posner M.R., Vermorken J.B., Cruz Hernandez J.J., Bourredjem A., Calais G., Paccagnella A., Hitt R., et al. Taxane-cisplatin-fluorouracil as induction chemotherapy in locally advanced head and neck cancers: an individual patient data meta-analysis of the meta-analysis of chemotherapy in head and neck cancer group. J. Clin. Oncol. 2013;31:2854–2860. doi: 10.1200/JCO.2012.47.7802. - DOI - PubMed
-
- Lefebvre J.L., Andry G., Chevalier D., Luboinski B., Collette L., Traissac L., de Raucourt D., Langendijk J.A., Head E, Neck Cancer G Laryngeal preservation with induction chemotherapy for hypopharyngeal squamous cell carcinoma: 10-year results of EORTC trial 24891. Ann. Oncol.: Off. J. Eur. Soc. Med. Oncol. /ESMO. 2012;23:2708–2714. doi: 10.1093/annonc/mds065. - DOI - PMC - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Research Materials

