Computational analysis of cas proteins unlocks new potential in HIV-1 targeted gene therapy
- PMID: 38239625
- PMCID: PMC10794619
- DOI: 10.3389/fgeed.2023.1248982
Computational analysis of cas proteins unlocks new potential in HIV-1 targeted gene therapy
Abstract
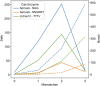
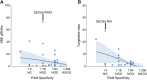
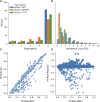
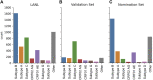
Introduction: The human immunodeficiency virus type 1 (HIV-1) pandemic has been slowed with the advent of anti-retroviral therapy (ART). However, ART is not a cure and as such has pushed the disease into a chronic infection. One potential cure strategy that has shown promise is the Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR)/Cas gene editing system. It has recently been shown to successfully edit and/or excise the integrated provirus from infected cells and inhibit HIV-1 in vitro, ex vivo, and in vivo. These studies have primarily been conducted with SpCas9 or SaCas9. However, additional Cas proteins are discovered regularly and modifications to these known proteins are being engineered. The alternative Cas molecules have different requirements for protospacer adjacent motifs (PAMs) which impact the possible targetable regions of HIV-1. Other modifications to the Cas protein or gRNA handle impact the tolerance for mismatches between gRNA and the target. While reducing off-target risk, this impacts the ability to fully account for HIV-1 genetic variability. Methods: This manuscript strives to examine these parameter choices using a computational approach for surveying the suitability of a Cas editor for HIV-1 gene editing. The Nominate, Diversify, Narrow, Filter (NDNF) pipeline measures the safety, broadness, and effectiveness of a pool of potential gRNAs for any PAM. This technique was used to evaluate 46 different potential Cas editors for their HIV therapeutic potential. Results: Our examination revealed that broader PAMs that improve the targeting potential of editors like SaCas9 and LbCas12a have larger pools of useful gRNAs, while broader PAMs reduced the pool of useful SpCas9 gRNAs yet increased the breadth of targetable locations. Investigation of the mismatch tolerance of Cas editors indicates a 2-missmatch tolerance is an ideal balance between on-target sensitivity and off-target specificity. Of all of the Cas editors examined, SpCas-NG and SPRY-Cas9 had the highest number of overall safe, broad, and effective gRNAs against HIV. Discussion: Currently, larger proteins and wider PAMs lead to better targeting capacity. This implies that research should either be targeted towards delivering longer payloads or towards increasing the breadth of currently available small Cas editors. With the discovery and adoption of additional Cas editors, it is important for researchers in the HIV-1 gene editing field to explore the wider world of Cas editors.
Keywords: CRiSPR/Cas; HIV-1; cure strategy; gene therapy; guideRNA.
Copyright © 2024 Dampier, Berman, Nonnemacher and Wigdahl.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



Similar articles
-
Gene Therapy with CRISPR/Cas9 Coming to Age for HIV Cure.AIDS Rev. 2017 Oct-Dec;19(3):167-172. AIDS Rev. 2017. PMID: 29019352
-
Developing Heritable Mutations in Arabidopsis thaliana Using a Modified CRISPR/Cas9 Toolkit Comprising PAM-Altered Cas9 Variants and gRNAs.Plant Cell Physiol. 2019 Oct 1;60(10):2255-2262. doi: 10.1093/pcp/pcz118. Plant Cell Physiol. 2019. PMID: 31198958
-
Designing broad-spectrum anti-HIV-1 gRNAs to target patient-derived variants.Sci Rep. 2017 Oct 31;7(1):14413. doi: 10.1038/s41598-017-12612-z. Sci Rep. 2017. PMID: 29089503 Free PMC article.
-
CRISPR-Cas nucleases and base editors for plant genome editing.aBIOTECH. 2019 Nov 30;1(1):74-87. doi: 10.1007/s42994-019-00010-0. eCollection 2020 Jan. aBIOTECH. 2019. PMID: 36305010 Free PMC article. Review.
-
Strategies for Optimization of the Clustered Regularly Interspaced Short Palindromic Repeat-Based Genome Editing System for Enhanced Editing Specificity.Hum Gene Ther. 2022 Apr;33(7-8):358-370. doi: 10.1089/hum.2021.283. Epub 2022 Mar 1. Hum Gene Ther. 2022. PMID: 34963339 Review.
Cited by
-
Delivering CRISPR to the HIV-1 reservoirs.Front Microbiol. 2024 May 15;15:1393974. doi: 10.3389/fmicb.2024.1393974. eCollection 2024. Front Microbiol. 2024. PMID: 38812680 Free PMC article. Review.
-
What's in a cure: designing a broad-spectrum HIV gene therapy.Curr Opin HIV AIDS. 2024 May 1;19(3):150-156. doi: 10.1097/COH.0000000000000846. Epub 2024 Mar 1. Curr Opin HIV AIDS. 2024. PMID: 38547339 Free PMC article. Review.
-
Impact of chromatin on HIV-1 latency: a multi-dimensional perspective.Epigenetics Chromatin. 2025 Mar 8;18(1):9. doi: 10.1186/s13072-025-00573-x. Epigenetics Chromatin. 2025. PMID: 40055755 Free PMC article. Review.
-
CRISPR/Cas9 for achieving postintervention HIV control.Curr Opin HIV AIDS. 2025 Sep 1;20(5):432-440. doi: 10.1097/COH.0000000000000963. Epub 2025 Jul 18. Curr Opin HIV AIDS. 2025. PMID: 40682401 Free PMC article. Review.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

