Blocking of microglia-astrocyte proinflammatory signaling is beneficial following stroke
- PMID: 38240014
- PMCID: PMC10794541
- DOI: 10.3389/fnmol.2023.1305949
Blocking of microglia-astrocyte proinflammatory signaling is beneficial following stroke
Abstract
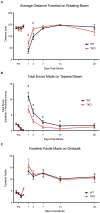
Microglia and astrocytes play an important role in the neuroinflammatory response and contribute to both the destruction of neighboring tissue as well as the resolution of inflammation following stroke. These reactive glial cells are highly heterogeneous at both the transcriptomic and functional level. Depending upon the stimulus, microglia and astrocytes mount a complex, and specific response composed of distinct microglial and astrocyte substates. These substates ultimately drive the landscape of the initiation and recovery from the adverse stimulus. In one state, inflammation- and damage-induced microglia release tumor necrosis factor (TNF), interleukin 1α (IL1α), and complement component 1q (C1q), together "TIC." This cocktail of cytokines drives astrocytes into a neurotoxic reactive astrocyte (nRA) substate. This nRA substate is associated with loss of many physiological astrocyte functions (e.g., synapse formation and maturation, phagocytosis, among others), as well as a gain-of-function release of neurotoxic long-chain fatty acids which kill neighboring cells. Here we report that transgenic removal of TIC led to reduction of gliosis, infarct expansion, and worsened functional deficits in the acute and delayed stages following stroke. Our results suggest that TIC cytokines, and likely nRAs play an important role that may maintain neuroinflammation and inhibit functional motor recovery after ischemic stroke. This is the first report that this paradigm is relevant in stroke and that therapies against nRAs may be a novel means to treat patients. Since nRAs are evolutionarily conserved from rodents to humans and present in multiple neurodegenerative diseases and injuries, further identification of mechanistic role of nRAs will lead to a better understanding of the neuroinflammatory response and the development of new therapies.
Keywords: astrocyte; infarct volume reduction; macrophage; recovery of function; stroke.
Copyright © 2024 Prescott, Münch, Brahms, Weigel, Inoue, Buckwalter, Liddelow and Peterson.
Conflict of interest statement
SL declares ownership in AstronauTx Ltd, and sits on the Scientific Advisory Board of the Global BioAccess Fund, Tamborine, and Synapticure. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
Figures



Similar articles
-
Blocking Formation of Neurotoxic Reactive Astrocytes is Beneficial Following Stroke.bioRxiv [Preprint]. 2023 Oct 16:2023.10.11.561918. doi: 10.1101/2023.10.11.561918. bioRxiv. 2023. PMID: 37905154 Free PMC article. Preprint.
-
M2 microglial small extracellular vesicles reduce glial scar formation via the miR-124/STAT3 pathway after ischemic stroke in mice.Theranostics. 2021 Jan 1;11(3):1232-1248. doi: 10.7150/thno.48761. eCollection 2021. Theranostics. 2021. PMID: 33391532 Free PMC article.
-
Cottonseed oil alleviates ischemic stroke injury by inhibiting the inflammatory activation of microglia and astrocyte.J Neuroinflammation. 2020 Sep 11;17(1):270. doi: 10.1186/s12974-020-01946-7. J Neuroinflammation. 2020. PMID: 32917229 Free PMC article.
-
Glial Cells: Role of the Immune Response in Ischemic Stroke.Front Immunol. 2020 Feb 26;11:294. doi: 10.3389/fimmu.2020.00294. eCollection 2020. Front Immunol. 2020. PMID: 32174916 Free PMC article. Review.
-
Activation of microglia and astrocytes: a roadway to neuroinflammation and Alzheimer's disease.Inflammopharmacology. 2019 Aug;27(4):663-677. doi: 10.1007/s10787-019-00580-x. Epub 2019 Mar 14. Inflammopharmacology. 2019. PMID: 30874945 Review.
Cited by
-
Acupuncture regulates astrocyte neurotoxic polarization to protect blood-brain barrier integrity in delayed thrombolysis through mediating ERK1/2/Cx43 axis.IBRO Neurosci Rep. 2025 Apr 11;18:604-618. doi: 10.1016/j.ibneur.2025.04.005. eCollection 2025 Jun. IBRO Neurosci Rep. 2025. PMID: 40292080 Free PMC article.
-
Changes in border-associated macrophages after stroke: Single-cell sequencing analysis.Neural Regen Res. 2026 Jan 1;21(1):346-356. doi: 10.4103/NRR.NRR-D-24-01092. Epub 2025 Jan 29. Neural Regen Res. 2026. PMID: 39927762 Free PMC article.
-
Updates of the role of B-cells in ischemic stroke.Front Cell Neurosci. 2024 Mar 14;18:1340756. doi: 10.3389/fncel.2024.1340756. eCollection 2024. Front Cell Neurosci. 2024. PMID: 38550918 Free PMC article. Review.
-
Aldh1l1-Cre/ERT2 Drives Flox-Mediated Recombination in Peripheral and CNS Infiltrating Immune Cells in Addition to Astrocytes During CNS Autoimmune Disease.Brain Behav. 2025 Feb;15(2):e70239. doi: 10.1002/brb3.70239. Brain Behav. 2025. PMID: 39910805 Free PMC article.
-
Neuronal LAMP2A-mediated reduction of adenylyl cyclases induces acute neurodegenerative responses and neuroinflammation after ischemic stroke.Cell Death Differ. 2025 Feb;32(2):337-352. doi: 10.1038/s41418-024-01389-0. Epub 2024 Sep 28. Cell Death Differ. 2025. PMID: 39341961
References
-
- Bates D., Kliegl R., Vasishth S., Baayen H. (2015). Parsimonious mixed models. arXiv [Preprint]. arXiv:1506.04967.
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

